Abstract
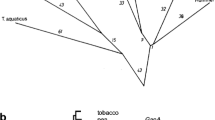
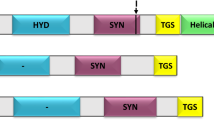
In a recent paper (Wenderoth et al., J Biol Chem 272: 26985–26990, 1997) we reported that the positions of the two redox regulatory cysteines identified in a plastidic G6PD isoform from potato (Solanum tuberosum L.) differ substantially from those conserved in cyanobacterial G6PDH sequences. To investigate the origin of redox regulation in G6PDH enzymes from photoautotrophic organisms, we isolated and characterized several G6PD cDNA sequences from higher plants and from a green and a red alga. Alignments of the deduced amino acid sequences showed that the cysteine residues cluster in the coenzyme-binding domain of the plastidic isoforms and are conserved at three out of six positions. Comparison of the mature proteins and the signal peptides revealed that two different plastidic G6PDH classes (P1 and P2) evolved from a common ancestral gene. The two algal sequences branch off prior to this class separation in higher plants, sharing about similar amino acid identity with either of the two plastidic G6PDH classes. The genes for cytosolic plant isoforms clearly share a common ancestor with animal and fungal G6PDH homologues, whereas the cyanobacterial isoforms branch within the eubacterial G6PDH sequences. The data suggest that cysteine-mediated redox regulation arose independently in G6PDH isoenzymes of eubacterial and eukaryotic lineages.
Similar content being viewed by others
References
Buchanan, B.B. 1991. Regulation of CO2 assimilation in oxigenic photosynthesis: the ferredoxin/thioredoxin system. Arch. Biochem. Biophys. 288: 1–9.
Bult, C.J., White, O., Olsen, G.J., Zhou, L., Fleischmann, R.D., Sutton, G.G., Blake, J.A., FitzGerald, L.M., Clayton, R.A., Gocayne, J.D., Kerlavage, A.R., Dougherty, B.A., Tomb, J.-F., Adams, M.D., Reich, C.I., Overbeek, R., Kirkness, E.F., Weinstock, K.G., Merrick, J.M., Glodek, A., Scott, J.L., Geoghagen, N.S.M., Weidman, J.F., Fuhrmann, J.L., Nguyen, D., Utterback, T.R., Kelley, J.M., Peterson, J.D., Sadow, P.W., Hanna, M.C., Cotton, M.D., Roberts, K.M., Hurst, M.A., Kaine, B.P., Borodovsky, M., Klenk, H.-P., Fraser, C.M., Smith, H.O., Woese, C.R. and Venter, J.C. 1986. Complete genome sequence of the methanogenic Archeon, Methanococcus jannaschii. Science 273: 1058–1073.
Church, G.M. and Gilbert, W. 1984. Genomic sequencing. Proc. Natl. Acad. Sci. USA 81: 1991–1995.
Copeland, L. and Turner, J.F. 1987. The regulation of glycolysis and the pentose-phosphate pathway. In: A. Marcus (Ed.), The Biochemistry of plants, vol. 11, Academic press, New York, pp. 107–125.
Cossar, J.D., Rowell, P. and Stewart, W.D.P. 1984. Thioredoxin as a modulator of glucose-6-phosphate dehydrogenase in a N2-fixing cyanobacterium. J. Gen. Microbiol. 130: 991–998.
de Boer, A.D. and Weisbeek, P.J. 1991. Chloroplast protein topogenesis: import, sorting and assembly. Biochim. Biophys. Acta 1071: 221–253.
Dubois, F., Brugière, N., Sangwan, R.S. and Hirel, B. 1996. Localisation of tobacco cytosolic glutamine synthetase enzymes and the corresponding transcripts shows organ-and cell-specific patterns of protein synthesis and gene expression. Plant Mol. Biol. 31: 803–817.
Elledge, S.J., Mulligan, J.T., Ramer, S.W., Spottswood, M. and Davis, R.W. 1991. λYES: a multifunctional cDNA expression vector for the isolation of genes by complementation of yeast and Escherichia coli mutations. Proc. Natl. Acad. Sci. USA 88: 1731–1735.
Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39: 783–791.
Fukasawa-Akada, T., Kung, S.D. and Watson, J.C. 1996. Phenylalanine ammonia-lyase gene structure, expression, and evolution in Nicotiana. Plant Mol. Biol. 30: 711–722.
Gavel, Y. and von Heijne, G. 1990. A conserved cleavage-site motif in chloroplast transit peptides. FEBS Lett. 261: 455–458.
Graeve, K., von Schaewen, A. and Scheibe, R. 1994. Purification, characterization and cDNA sequence of glucose-6-phosphate dehydrogenase from potato (Solanum tuberosum L.). Plant J. 5: 353–361.
Kawarabayasi, Y., Sawada, M., Horikawa, H., Haikawa, Y., Hino, Y., Yamamoto, S., Sekine, M., Baba, S.-I., Kosugi, H., Hosoyama, A., Nagai, Y., Sakai, M., Ogura, K., Otsuka, R., Nakazawa, H., Takamiya, M., Ohfuku, Y., Funahashi, T., Tanaka, T., Kudoh, Y., Yamazaki, J., Kushida, N., Oguchi, A., Aoki, K.-I., Yoshizawa, T., Nakamura, Y., Robb, F.T., Horikoshi, K., Masuchi, Y., Shizuya, H. and Kikuchi, H. 1998. Complete sequence and gene organization of the genome of a hyper-thermophilic Archaebacterium, Pyrococcus horikoshii OT3 (supplement). DNA Res. 5 (Suppl.): 147–155.
Klenk, H.P., Clayton, R.A., Tomb, J.F., White, O., Nelson, K.E., Ketchum, K.A., Dodson, R.J., Gwinn, M., Hickey, E.K., Peterson, J.D., Richardson, D.L., Kerlavage, A.R., Graham, D.E., Kyrpides, N.C., Fleischmann, R.D., Quackenbush, J., Lee, N.H., Sutton, G.G., Gill, S., Kirkness, E.F., Dougherty, B.A., Mckenney, K., Adams, M.D., Loftus, B., Peterson, S., Reich, C.I., McNeil, L.K., Badger, J.H., Glodek, A., Zhou, L., Overbeek, R., Gocayne, J.D., Weidman, J.F., McDonald, L., Utterback, T., Cotton, M.D., Spriggs, T., Artiach, P., Kaine, B.P., Sykes, S.M., Sadow, P.W., D'Andrea, K.P., Bowman, C., Fujii, C., Garland, S.A., Mason, T.M., Olsen, G.J., Fraser, C.M., Smith, H.O., Woese, C.R. and Venter, J.C. 1997. The complete genome sequence of the thermophilic, sulphate-reducing Archaeon Archaeoglobus fulgidus. Nature 390: 364–370.
Knight, M.E., Halpin, C. and Schuch, W. 1992. Identification and characterisation of cDNA clones encoding cinnamyl alcohol dehydrogenase from tobacco. Plant Mol. Biol. 19: 793–801.
Kronenberger, J., Lepingle, A., Caboche, M. and Vaucheret, H. 1993. Cloning and expression of distinct nitrite reductases in tobacco leaves and roots. Mol. Gen. Genet. 236: 203–208.
Lechtreck, K.-F., Frins, S., Bilski, J., Teltenkötter, A., Weber, K. and Melkonian, M. 1996. The cruciated microtubule-associated fibers of the green alga Dunaliella bioculata consist of a 31 kDa SF-assemblin. J. Cell Sci. 109: 827–835.
Lee, D. and Douglas, C.J. 1996. Two different members of a tobacco 4-coumarate:coenzyme A ligase (4CL) gene family. Plant Physiol. 112: 193–205.
Lee, K.Y., Townsend, J., Tepperman, J., Black, M., Chui, C.F., Mazur, B., Dunsmuir, P. and Bedbrook, J. 1988. The molecular basis of sulfonylurea herbicide resistance in tobacco. EMBO J. 1241–1248.
Martin,W. and Herrmann, R.G. 1998. Gene transfer from organelles to the nucleus: how much, what happens, and why? Plant Physiol. 118: 9–17.
Martin, W. and Schnarrenberger, C. 1997. The evolution of the Calvin cycle from prokaryotic to eukaryotic chromosomes: a case study of functional redundancy in ancient pathways through endosymbiosis. Curr. Genet. 32: 1–18.
Narayan, R.K.J. 1987. Nuclear DNA changes, genome differentiation and evolution in Nicotiana (Solanaceae). Plant Syst. Evol. 157: 161–180.
Newman, J., Karakaya, H., Scanlan, D.J. and Mann, N.H. 1995. A comparison of gene organization in the zwf region of the genomes of the cyanobacteria Synechococcus sp. PCC 7942 and Anabaena sp. PCC 7120. FEMS Microbiol. Lett. 133: 187–193.
Nicholas, K.B. and Nicholas, H.B. 1997. GeneDoc: a tool for editing and annotating multiple sequence alignments. Distributed by the authors.
Page, R.D.M. 1996. TREEVIEW: an application to display phylogenetic trees on personal computers. Comp. Appl. Biosci. 12: 357–358.
Purwantini, E., Gillis, T.P. and Daniels, L. 1997. Presence of F420-dependent glucose-6-phosphate dehydrogenase in Mycobacterium and Nocardia species, but absence from Streptomyces and Corynebacterium species and methanogenic Archaea. FEMS Microbiol. Lett. 146: 129–134.
Rossmann, M.G., Liljas, A., Brändén, C.I. and Banaszak, L.J. 1975. Evolutionary and structural relationship among dehydrogenases. In: P.D. Boyer (Ed.), The Enzymes, 11, Academic Press, New York, pp. 61–102.
Saitou, N. and Nei, M. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4: 406–425.
Sambrook, J., Fritsch, E.F. and Maniatis, P.J. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
Scanlan, D.J., Newman, J., Sebaihia, M., Mann, N.H. and Carr, N.G. 1992. Cloning and sequence analysis of the glucose-6-phosphate dehydrogenase gene from the cyanobacterium Synechococcus PCC 7942. Plant Mol. Biol. 19: 877–880.
Scheibe, R. 1990. Light/dark modulation: regulation of chloroplast metabolism in a new light. Bot. Acta 103: 327–334.
Smith, D.R., Doucette-Stamm, L.A., Deloughery, C., Lee, H.M., Dubois, J., Aldredge, T., Bashirzadeh, R., Blakely, D., Cook, R., Gilbert, K., Harrison, D., Hoang, L., Keagle, P., Lumm, W., Pothier, B., Qiu, D.Y., Spadafora, R., Vicaire, R., Wang, Y., Wierzbowski, J., Gibson, R., Jiwani, N., Caruso, A., Bush, D., Safer, H., Patwell, D., Prabhakar, S., McDougall, S., Shimer, G., Goyal, A., Pietrokovski, S., Church, G.M., Daniels, C.J., Mao, J.-I., Rice, P., Nölling, J. and Reeve, J.N. 1997. Complete genome sequence of Methanobacterium thermoautotrophicum 1H: Functional analysis and comparative genomics. J. Bact. 179: 7135–7155.
Summers, M.L., Meeks, J.C., Chu, S. and Wolf, R.E. Jr. 1995. Plant Gene register. Nucleotide sequence of an operon in Nostoc sp. strain ATCC 29133 encoding four genes of the oxidative pentose phosphate cycle. Plant Physiol. 107: 267–268.
Thompson, J.D., Higgins, D.G. and Gibson, T.J. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucl. Acids Res. 22: 4673–4680.
van Buuren, M., Neuhaus, J.-M., Shinshi, H., Ryals, J. and Meins, F. 1992. The structure and regulation of homologous tobacco endochitinase genes of Nicotiana sylvestris and N. tomentosiformis origin. Biochem. Plants 2: 279–316.
Vaucheret, H., Vincenentz, M., Kronenberger, J., Caboche, M. and Rouzé, P. 1989. Molecular cloning and characterisation of the two homologous genes coding for nitrate reductase in tobacco. Mol. Gen. Genet. 232: 460–469.
von Schaewen, A., Langenkämper, G., Graeve, K., Wenderoth, I. and Scheibe, R. 1995. Molecular characterization of the plastidic glucose-6-phosphate dehydrogenase from potato in comparison to its cytosolic counterpart. Plant Physiol. 109: 1327–1335.
Wenderoth, I., Scheibe, R. and von Schaewen, A. 1997. Identifi-cation of the cysteine residues involved in redox modification of plant plastidic glucose-6-phosphate dehydrogenase. J. Biol. Chem. 272: 26985–26990.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wendt, U.K., Hauschild, R., Lange, C. et al. Evidence for functional convergence of redox regulation in G6PDH isoforms of cyanobacteria and higher plants. Plant Mol Biol 40, 487–494 (1999). https://doi.org/10.1023/A:1006257230779
Issue Date:
DOI: https://doi.org/10.1023/A:1006257230779