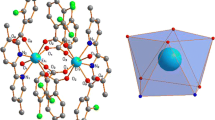
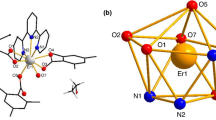
Abstract
Thermal behaviour of a few lanthanide complexes of the type ML3(I) [M=Eu,Gd; HL=4,4,4-trifluoro- 1-(2-napthyl)-1,3-butanedione and EuL30.5dmm dmm=2,6-dimethylmorpholine(II)], has been investigated. From thermogravimetric(TG) curves, the decomposition pattern of the compounds has been analysed on the basis of mass loss data. The order and activation energy of the thermal decomposition reactions have been elucidated. From differential thermal analysis (DTA) studies, the heat of reaction and rate of thermal decomposition reaction have been enumerated.
Similar content being viewed by others
References
A. K. Trikha, A. Kumar and S. Kaur, Ann. Braz. Chem. Assoc., 44 (1995) 23.
A. K. Trikha, S. Kaur, L. B. Zineer, K. Zinner and P. C. Isolani, 35th Congress Brasileiro de Quimica, 1995.
A. K. Trikha, A. Kumar and S. Kaur, J. Fluorine Chem., 78 (1996) 109.
A. K. Trikha and G. S. Sodhi, J. Thermal Anal., 41 (1994) 909.
A. W. Coats and J. P. Redfern, Nature (London), 201 (1964) 68.
W. E. Collins, in ‘Analytical Calorimetry’, Vol. 2, R. S. Porter and J. M. Johnson (Eds.), Plenum, New York 1970, p. 353.
B. R. Currell, in ‘Thermal Analysis’, Vol. 3, R. F. Schwenker and P. D. Garn (Eds.), Academic Press, New York 1969, p. 1185.
H. J. Borchardt, J. Inorg. Nucl. Chem., 12 (1960) 252.