Abstract
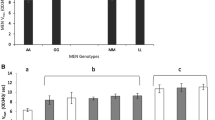
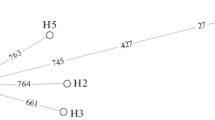
Six naturally occurring but rare alleles of sn-glycerol-3-phosphate dehydrogenase (Gpdh) in Drosophila melanogaster have been investigated in this study. They all belong to a class of Gpdh UF (ultra-fast) alleles, because their electrophoretic mobilities are faster than that of the Gpdh F (fast) allele. The Gpdh UF variants are widespread, and have been reported from five continents. DNA sequence analysis has shown that the change in electrophoretic mobility was in each allele caused by a single amino acid residue substitution in the encoded protein. In the Xiamen UF allele it is a substitution of lysine (AAA) to asparagine (AAT) in exon 1 (residue 3). An asparagine (AAT) to aspartate (GAT) change was found in exon 6 (residue 336) in the Iowa UF and Netherlands UF alleles. The mobility of the Raleigh UF allele was altered by a valine (GTG) to glutamate (GAG) substitution in exon 3 (residue 76). Two mutations were detected in the Brazzaville UF allele: a lysine (AAG) to methionine (ATG) substitution in exon 2 (residue 68) is responsible for the ultra-fast phenotype of this variant, while a tyrosine (TAT) to phenylalanine (TTT) substitution in exon 4 (residue 244) is not expected to alter the electrophoretic mobility of the encoded protein. These results indicate that the Gpdh UF alleles originate from different mutational events, and only two of them — Iowa UF and Netherlands UF — might share a common ancestry. The GPDH activity of the Iowa UF allele is intermediate between those of the Gpdh S and Gpdh F control stocks. The other Gpdh UF variants have lower activities than the controls: Xiamen UF-83%, Raleigh UF-80% and Brazzaville UF-73% of the Gpdh F control.
Similar content being viewed by others
References
Barton, N.H. & M. Slatkin, 1986. A quasi-equilibrium theory of the distribution of rare alleles in a subdivided population. Heredity 56: 409–415.
Bewley, G.C., D.W. Niesel & J.R. Wilkins, 1984. Purification and characterization of the naturally occurring allelic variants of sn-glycerol-3-phosphate dehydrogenase in Drosophila melanogaster, Comp. Biochem. Physiol. 79B: 23–32.
Bewley, G.C., J.L. Cook, S. Kusakabe, T. Mukai, D.L. Rigby & G.K. Chambers, 1989. Sequence, structure and evolution of the gene coding for sn-glycerol-3-phosphate dehydrogenase in Drosophila melanogaster. Nucl. Acids Res. 17: 8553–8567.
Charles-Palabost, L., 1989. Alleles found at six gene-enzyme systems in the French natural populations of Drosophila melanogaster, DIS 63: 43–44.
Chia, W., C. Savakis, R. Karp, H. Pelham & M. Ashburner, 1985. Mutation of the Adh gene of Drosophila melanogaster containing an internal tandem duplication. J. Mol. Biol. 186: 679–688.
David, J.R., 1982. Latitudinal variability of Drosophila melanogaster: allozyme frequencies divergence between european and afrotropical populations. Biochem. Genet. 20: 747–761.
Devereux, J., P. Haeberli & O. Smithies, 1984. A comprehensive set of sequence analysis programs for the VAX. Nucl. Acids Res. 12: 387–395.
Dobzhansky, T., 1973. Active dispersal and passive transport in Drosophila. Evolution 27: 565–575.
El-Abidin Salam, A.Z., A.M. El-Adl & Z.A. Kosba, 1982. Isozyme polymorphism in Drosophila. IV. Interspecific variation and population dynamics, pp. 227–235 in Advances in genetics, development and evolution of Drosophila, edited by S. Lakovaara, Plenum Press, New York, USA.
Gibson, J.B., A.V. Wilks, A. Cao & A.L. Freeth, 1986. Dominance for sn-glycerol-3-phosphate dehydrogenase activity in Drosophila melanogaster: evidence for differential allelic expression mediated via a trans-acting effect. Heredity 56: 227–235.
Gibson, J.B., A. Cao, J. Symonds & D. Reed, 1991. Low activity sn-glycerol-3-phosphate dehydrogenase variants in natural populations of Drosophila melanogaster. Heredity 66: 75–82.
Grell, E.H., 1967. Electrophoretic variants of α-glycerophosphate dehydrogenase in Drosophila melanogaster. Science 158: 1319–1320.
Jiang, C., J.B. Gibson & H. Chen, 1989. Genetic differentiation in populations of Drosophila melanogaster from the People's Republic of China: comparison with patterns on other continents. Heredity 62: 193–198.
Kamping, A. & W.van Delden, 1988. Hybridization between D. melanogaster and D. simulans in nature. DIS 67: 53.
Kamping, A. & W.van Delden, 1991. Biochemical differences between α-GPDH genotypes in Drosophila melanogaster. DIS 70: 117–118.
Lee, C.-Y., D. Niesel & G.C. Bewley, 1980. Analyses of genetic variants of L-glycerol-3-phosphate dehydrogenase in Drosophila melanogaster by two-dimensional gel electrophoresis and immunoelectrophoresis. Biochem. Genet. 18: 1003–1018.
Lodi, P.J. & J.R. Knowles, 1991. Neutral imidazole is the electrophile in the reaction catalyzed by triosephosphate isomerase: structural origins and catalytic implications. Biochemistry 30: 6948–6956.
Niesel, D.W., Y.-C.E. Pan, G.C. Bewley, F.B. Armstrong & S.S.-L. Li, 1982. Structural analysis of adult and larval isozymes of sn-glycerol-3-phosphate dehydrogenase of Drosophila melanogaster. J. Biol. Chem. 257: 979–983.
Oakeshott, J.G., J.B. Gibson, P.R. Anderson, W.R. Knibb, D.G. Anderson & G.K. Chambers, 1982. Alcohol dehydrogenase and glycerol-3-phosphate dehydrogenase clines in Drosophila melanogaster on different continents. Evolution 36: 86–96.
Otto, J., P. Argos & M.G. Rossmann, 1980. Prediction of secondary structural elements in glycerol-3-phosphate dehydrogenase by comparison with other dehydrogenases. Eur. J. Biochem. 109: 325–330.
Reed, D.S. & J.B. Gibson, 1993. Defective P element insertions affect the expression of sn-glycerol-3-phosphate dehydrogenase alleles in natural populations of Drosophila melanogaster. Proc. R. Soc. Lond. B 251: 39–45.
Reed, D.S. & J.B. Gibson, 1994. Molecular heterogeneity of naturally occurring sn-glycerol-3-phosphate dehydrogenase low-activity variants in Drosophila melanogaster. Biochem. Genet. 32: 161–179.
Singh, R.S. & L.R. Rhomberg, 1987. A comprehensive study of genic variation in natural populations of Drosophila melanogaster. I. Estimates of gene flow from rare alleles. Genetics 115: 313–322.
Slatkin, M., 1985. Rare alleles as indicators of gene flow. Evolution 39: 53–65.
Smith, J.M. & J. Haigh, 1974. The hitch-hiking effect of a favourable gene. Genet. Res. 23: 23–35.
Symonds, J.E. & J.B. Gibson, 1992. Restriction site variation, gene duplication, and the activity of sn-glycerol-3-phosphate dehydrogenase in Drosophila melanogaster. Biochem. Genet. 30: 169–188.
van Delden, W. & A. Kamping, 1988. Test for viability differences among α-Gpdh genotypes in D. melanogaster. DIS 67: 87.
von Kalm, L., J. Weaver, J. DeMarco, R.J. MacIntyre & D.T. Sullivan, 1989. Structural characterization of the α-glycerol-3-phosphate dehydrogenase-encoding gene of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 86: 5020–5024.
Woodruff, R.C. & M. Ashburner, 1979. The genetics of a small autosomal region of Drosophila melanogaster containing the structural gene for alcohol dehydrogenase. I. Characterization of deficiencies and mapping of Adh and visible mutations. Genetics 92: 117–132.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wilanowski, T.M., Barnes, P.T. & Gibson, J.B. Molecular structure of rare but geographically widespread sn-glycerol-3-phosphate dehydrogenase ‘ultra-fast’ electrophoretic alleles in Drosophila melanogaster . Genetica 97, 165–172 (1996). https://doi.org/10.1007/BF00054623
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00054623