Abstract
Inputs of surface waters high in chloride and sulphateincreased the availability of nutrients in fenpeatlands. This `internal eutrophication' wasdemonstrated with test plants (`phytometers') andthrough water and soil analysis. Three experiments arepresented in which the level of chloride and/orsulphate was increased to 3 mmolc l−1. Inexperiment 1 chloride levels were increased from 0.5to 3 mmolc l−1 as CaCl2 or NaCl. Inexperiment 2 and 3 similar increased levels forchloride and sulphate (3 mmolc l−1; as NaCland Na2SO4) were used. The following resultswere found:
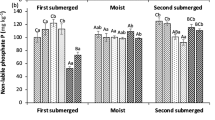
(i) No differences in soil total-N and total-P werefound before and after the treatments in any of thethree experiments.
(ii) Experiment 1 showed a significant increase inBio-Available P (BAP) in pots planted with Anthoxanthum odoratum as well as in bare pots for theNaCl and CaCl2 treatments. The plants in thesetreatments had taken up much more P.
(iii) Experiment 2 showed an increase in soil BAPafter treatment with chloride and sulphate in potsplanted with Anthoxanthum odoratum. The chloridetreatment had no effect on plant biomass, whereas thesulphate treatment resulted in a reduction in rootbiomass and root N and P content. The shoots showedan increase in P content in the sulphate and chloridetreatments, while N content remained the same.
(iv) In experiment 3, treatments with chloride andsulphate led to significantly increased biomass and Puptake of Anthoxanthum odoratum. Again, noeffects on N uptake were found.
These experiments provide evidence for distinctlyincreased availability of phosphate in peat soils whenthese come into contact with water with evenmoderately increased sulphate or chloride levels.Surface water originating from the Rhine river, whichis enriched in chloride and sulphate, is oftensupplied to fen reserves in The Netherlands, tocompensate for water losses due to agriculturaldrainage in the region. The results of this study showthat phosphate availability to the vegetation may risedrastically, with detrimental effects on the speciesdensity and the occurrence of rare species in thevegetation. Hence, supply of this water should beavoided.
Similar content being viewed by others
References
Aerts R & Ludwig F (1997) Water-table changes and nutritional status affect trace gas emissions from laboratory columns of peatland soils. Soil Biol. Biochem. 29: 1691–1699
Appelo CAJ & Postma D (1993) Geochemistry, Groundwater and Pollution. Balkema, Rotterdam
Beltman B (1990) Aquatic Macrophytes: A Useful Tool Against Eutrophication. 8th Int. Symp. on Aquat. Weeds (pp 35–38). Eur. Weed Res. Soc.
Beltman B, Kooijman AJM, Ellers J & Oosterbeek BJ (1992) Nutrient availability and plant species composition of rich fens in the dune complex at Dooaghtry, Co. Mayo, Ireland. In: Carter RWG, Curtis TGF & Sheehy-Skeffington E (Ed) Ecology, Geomorphology and Management of Coastal Dunes (pp 283–296). North-Holland Publ., Amsterdam
Boyer MLH & Wheeler BD (1989)Vegetation patterns in spring-fed calcarous fens: calcite precipitation and constraints on fertility. J. Ecol. 77: 597–609
Caraco NF, Cole JJ & Likens GE (1989) Evidence for sulphate-controlled phosphorus release from sediment of aquatic systems. Nature 341: 316–318
Curtis PJ (1989) Effects of hydrogen ion and sulphate on the phosphorus cycle of a precambian Shield Lake. Nature 337: 156–158
Fixen PE, Ludwick AE & Olsen SR (1983) Phosphorus and potassium fertilization of irrigated alfafa on calcareous soils. II Soil phosphorus solubility relationships. Soil Sci. Soc. Am. J. 47: 112–117
Freeze RA & Cherry JA (1979) Groundwater. Prentice-Hall, Englewood Cliffs, NJ
Geelhoed JS, Hiemstra T & Van Riemsdijk WH (1997a) Phosphate and sulphate adsorption on goethite: Single anion and compatitive adsorption. Geochim. Cosmoch. 61: 2389–2396
Geelhoed JS, Van Riemsdijk WH & Findenegg GR (1997b) Effects of sulphate and pH on the plant-availability of phosphate adsorbed on goethite. Plant and Soil 197: 241–249
Heathwaite AL (1995) Overview of the hydrology of British Wetlands. In: Hughes JMR & Heathwaite AL (Ed) Hydrology and Hydrochemistry of British Wetlands (pp 11–21).J. Wiley, Chichester, England
Houba VJG, Van Schouwenburg JCh, Walinga I & Novozamsky J (1979) Soil Analysis. Agricultural University, Wageningen
Inskeep WP & Silvertooth JC (1988) Inhibition of hydroxyapatite precipitation in the presence of fulvic, humic and tannic soils. Soil. Sci. Soc. Am. J. 52: 941–946
Koerselman W (1989) Hydrology and nutrient budgets of fens in an agricultural landscape. PhD Thesis, Utrecht University
Koerselman W, Van Kerkhoven MB & Verhoeven JTA (1994) Release of inorganic N, P and K in peat soils; effects of temperature, water chemistry and water level. Biogeochem. 20: 644–647
Lamers LPM, Smolders AJP, Brouwer E & Roelofs JGM (1996) Is sulphate enriched water suitable for water inlet? The role of water quality in measures compensating for water shortage. Landschap 13: 19–30
Lamers LPM, Tomassen HBM & Roelofs JGM (1998) Sulphate-induced eutrophication and phytotoxicity in freshwater wetlands. Environ. Sci. Technol. 32: 199–205
Larsen S (1966) The solubility of phosphate in a calcareous soil.J. Soil Sci. 17: 121–127
O'Conner GA, Knudtsen KL & Connell GA (1986) Phosphorus solubility in sludge-amended calcareous soils. J. Envir. Qual. 15: 308–312
Perez Corona ME, Van der Klundert I & Verhoeven JTA (1996) Availability of organic and inorganic phosphorus compounds as phosphorus sources for Carex species.New Phytol. 133: 225–231
Piek H (1996) Personal communication
Qualls RG & Richardson CJ (1995) Forms of soil phosphorus along a nutrient enrichment gradient in the Northern Everglades. Soil Sci. 160: 183–198
Richardson CJ & Marshall PE (1986) Processes controlling movement, storage and export of phosphorus in a fen peatland. Ecol. Monographs65: 279–302
Richardson CJ (1989) Freshwater wetlands: transformers, filters or sinks. In: Sharitz, R.R. & J.W. Gibbons (Eds) Freshwater Wetlands and Wildlife, 1989, CONF-8603101. DOS Symposium Series no. 61, USDOS Office of Scientific and technical Information, Oak Ridge, TN
Roelofs JGM (1991) Inlet of alkaline river water into peaty lowlands: Effects on water quality and Stratiotes aloides stands. Aq. Bot. 39: 267–293
SAS (1988)SAS/STAT Users Guide for Personel Computers. SAS-inst. Inc., N. Carolina, U.S.A.
Smolders A & Roelofs JGM (1995) Internal eutrophication, iron limitation and sulfide accumulation due to the inlet of river Rhine water in peaty shallow waters in The Netherlands. Archiv f. Hydrobiol. 133: 349–365
Tipping E & Hurley MA (1988) A model of solid-solution interactions in acid organic soils, based on the complexation properties of humic substances. J. Soil Sci. 39: 505–519
Tipping E, Backes CA & Hurley MA (1988) The complexation of protons, aluminium and calcium by aquatic humic substances: a model incorporating binding-site heterogeneity and macroionic effects. Wat. Res. 22: 597–611
Verhoeven JTA, Koerselman W & Beltman B (1988) The vegetation of fens in relation to their hydrology and nutrient dynamics. In: Symoens JJ (Ed) Vegetation of Inland Waters (pp 249–282). Kluwer Acad. Publ., Dordrecht
Verhoeven JTA, Kooijman AM & Van Wirdum G (1988) Mineralisation of N and P along a trophic gradient in a fresh water mire. Biogeochem. 6: 31–43
Verhoeven JTA, Maltby E & Schmitz MB (1990) Nitrogen and phosphorus mineralization in fens and bogs. J. Ecol. 78: 713–726
Verhoeven JTA (Ed) (1992) Fens and Bogs in the Netherlands: Vegetation, History, Nutrient Dynamics and Conservation. Geobotany 18, Kluwer Acad. Publ., Dordrecht
Wheeler BD (1980) Plant communities of rich-fen systems in England and Wales II: communities of calcarous mires. J. Ecol. 68: 405–420
Wheeler BD, Shaw SC & Cook RED (1992) Phytometric assessment of the fertility of undrained rich-fen soils. J. Appl. Ecol. 29: 466–475
Rights and permissions
About this article
Cite this article
Beltman, B., Rouwenhorst, T., Van Kerkhoven, M. et al. Internal eutrophication in peat soils through competition between chloride and sulphate with phosphate for binding sites. Biogeochemistry 50, 183–194 (2000). https://doi.org/10.1023/A:1006374018558
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1006374018558