Abstract
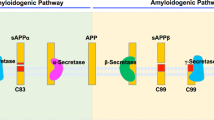
Cleavage of amyloid precursor protein (APP) by the β- and γ-secretases generates the amino and carboxy termini, respectively, of the Aβ amyloidogenic peptides Aβ40 and Aβ42—the major constituents of the amyloid plaques in the brain parenchyma of Alzheimer's disease patients1. There is evidence that the polytopic membrane-spanning proteins, presenilin 1 and 2 (PS1 and PS2), are important determinants of γ-secretase activity: mutations in PS1 and PS2 that are associated with early-onset familial Alzheimer's disease2,3 increase the production of Aβ42 (refs 4,5,6), the more amyloidogenic peptide; γ-secretase activity is reduced in neuronal cultures derived from PS1-deficient mouse embryos7; and directed mutagenesis of two conserved aspartates in transmembrane segments of PS1 inactivates the ability of γ-secretase to catalyse processing of APP within its transmembrane domain8. It is unknown, however, whether PS1 (which has little or no homology to any known aspartyl protease) is itself a transmembrane aspartyl protease or a γ-secretase cofactor, or helps to colocalize γ-secretase and APP. Here we report photoaffinity labelling of PS1 (and PS2) by potent γ-secretase inhibitors that were designed to function as transition state analogue inhibitors directed to the active site of an aspartyl protease. This observation indicates that PS1 (and PS2) may contain the active site of γ-secretase. Interestingly, the intact, single-chain form of wild-type PS1 is not labelled by an active-site-directed photoaffinity probe, suggesting that intact wild-type PS1 may be an aspartyl protease zymogen.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Selkoe, D. J. The cell biology of β-amyloid precursor protein and presenilin in Alzheimer's disease. Trends Cell Biol. 8, 447– 453 (1998).
Sherrington, R. et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer's disease. Nature 375, 754–760 (1995).
Levy-Lahad, E. et al. A familial Alzheimer's disease locus on chromosome 1. Science 269, 970–973 ( 1995).
Scheuner, D. et al. Secreted amyloid beta-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2, 864–870 ( 1996).
Duff, K. et al. Increased amyloid-beta42(43) in brains of mice expressing mutant presenilin 1. Nature 383, 710– 713 (1996).
Borchelt, D. R. et al. Familial Alzheimer's disease-linked presenilin 1 variants elevate Abetal-42,1-40 ratio in vitro and in vivo. Neuron 17, 1005–1013 (1996).
De Strooper, B. et al. Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391, 387 –390 (1998).
Wolfe, M. S. et al. Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and γ-secretase activity. Nature 398, 513–517 ( 1999).
Shearman, M. S. et al. L-685,458, an aspartic protease transition state mimic, is a potent inhibitor of APP γ-secretase activity. Biochemistry , in the press.
Wolfenden, R. Analog approaches to the structure of the transition state in enzyme reactions. Accts. Chem. Res. 5, 10– 18 (1972).
Rich, D. H. Inhibitors of Aspartic Proteinases. (eds. Barrett, A. J. & Salvesen, G.) 179–217 (Elsevier Science, 1986).
Li, Y.-M. et al. Presenilin-1 is linked with γ-secretase activity in the detergent solubilized state. Proc. Natl Acad. Sci. USA (in the press).
Wolfe, M. S. et al. Peptidomimetic probes and molecular modeling suggest that Alzheimer's γ-secretase is an intramembrane-cleaving aspartyl protease. Biochemistry 38, 4720– 4727 (1999).
Thinakaran, G. et al. Endoproteolysis of presenilin-1 and accumulation of processed derivatives in vivo. Neuron 17, 181– 190 (1996).
Seeger, M. et al. Evidence for phosphorylation of oligomeric assembly of presenilin 1. Proc. Natl Acad. Sci. USA 94, 5090– 5094 (1997).
Capell, A. et al. The proteolytic fragments of the Alzheimer's disease-associated presenilin-1 form heterodimers and occur as a 100–150-kDa molecular mass complex. J. Biol. Chem. 273, 3205– 3211 (1998).
Perez-Tur, J. et al. A mutation in Alzheimer's disease destroying a splice acceptor site in the presenilin-1 gene. Neuroreport 7, 297–301 (1995).
Thinakaran, G. et al. Evidence that levels of presenilins (PS1 and PS2) are coordinately regulated by competition for limiting cellular factors. J. Biol. Chem. 272, 28415–28422 ( 1997).
Evans, B. E. et al. A stereocontrolled synthesis of hydroxyethylene dipeptide isosteres using novel, chiral aminoalkyl epoxides and γ-(aminoalkyl) γ-lactones. J. Org. Chem. 50, 4615– 4625 (1985).
Yang, H., Leland, J. K., Yost, D. & Massey, R. J. Electrochemiluminescence: A new diagnostic and research tool. Bio/Technology 12, 193–194 (1994).
Khorkova, O. E., Patel, K., Heroux, J. & Sahasrabudhe, S. Modulation of amyloid precursor protein processing by compounds with various mechanisms of action: detection by liquid phase electrochemiluminescent system. J. Neurosci. Methods 82, 159–166 (1998).
Ida, N. et al. Analysis of heterogeneous βA4 peptides in human cerebrospinal fluid and blood by a newly developed sensitive western blot analysis. J. Biol. Chem. 271, 22908–22914 (1996).
Barelli, H. L. et al. Characterization of new polyclonal antibodies specific for 40 and 42 amino acid-long amyloid β-peptides: Their use to examine the cell biology of presenilins and the immunohistochemistry of sporadic Alzheimer's disease and cerebral amyloid angiopathy cases. Mol. Med. 3, 695–707 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, YM., Xu, M., Lai, MT. et al. Photoactivated γ-secretase inhibitors directed to the active site covalently label presenilin 1. Nature 405, 689–694 (2000). https://doi.org/10.1038/35015085
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35015085
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.