Abstract
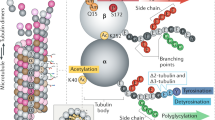
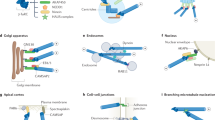
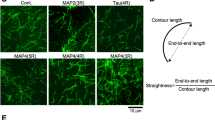
Microtubules are a key cytoskeletal constituent of the eukaryotic cell, being involved in mitosis, cell division, cell shape, intracellular transport and motility1. They are composed of tubulin, a 110,000-molecular weight (Mr) protein which is a dimer of 55,000-Mr α and β submits2. Multiple forms of both α-and β- tubulin have been demonstrated biochemically by gel electrophoresis3,4, column chromatography5 and peptide maps6; immunologically by monoclonal antibodies7; and genetically by mapping multiple tubulin genes in the eukaryotic genome8–10. There is evidence for multiple tubulin mRNA species11,12,13, but some of the microheterogeneity may also arise from posttranslational modifications such as phosphorylation14 and glycosylation15. Tubulin microheterogeneity is most dramatic in the brain where it could be the result of cellular heterogeneity or a multiplicity of specific functions within each cell, such as neurite extension and axoplasmic transport1,16. Functional specificity has been demonstrated in particular tubulin subunits in other tissues9,16,17. To determine whether multiple forms of tubulin coexist in single cells, we have purified and characterized the tubulin of single, isolated sympathetic neurones grown in primary cell culture18. We show here for the first time that tubulin can be isolated and characterized from a single mammalian neurone. Single neurones exhibit multiple tubulin isoelectric forms, suggesting that structurally different forms of microtubules have functionally different roles in a single cell.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dustin, P. Microtubules (Springer, New York, 1978).
Luduena, R. F., Shooter, E. M. & Wilson, L. J. biol. Chem. 252, 7006–7014 (1977).
Gozes, I. & Littauer, U. Z. Nature 276, 411–413 (1978).
Marotta, C. A., Harris, J. L. & Gilbert, J. M. J. Neurochem. 30, 1431–1440 (1978).
Lu, R. C. & Elzina, M. Analyt. Biochem. 77, 243–250 (1977).
Stephens, R. E. Biochemistry. 17, 2882–2891 (1978).
Gozes, I. & Barnstable, C. J. Proc. natn. Acad. Sci. U.S.A. (in the press).
Cleveland, D. W. et al. Cell. 20, 95–105 (1980).
Kemphues, K. J., Raff, E. C., Raff, R. A. & Kaufman, T. C. Cell 21, 445–451 (1980).
Silflow, C. D. & Rosenbaum, J. L. Cell 24, 81–88 (1981).
Gozes, I., de Baetselier, A. & Littauer, U. Z. Eur. J. Biochem. 103, 13–20 (1980).
Bryan, R. N., Bossinger, J. & Hayashi, M. Devl Biol. 81, 349–355 (1981).
Strocchi, P., Marotta, C. A., Bonventre, J. & Gilbert, J. M. Brain Res. 211, 206–210 (1981).
Forgue, S. T. & Dahl, J. L. J. Neurochem. 32, 1015–1025 (1979).
Feit, H. & Shelanski, M. L. Biochem. biophys. Res. Commun. 66, 920–927 (1975).
Gozes, I. Neurochem. Int. (in the press).
Cabral, F., Sobel, M. E. & Gottesman, M. M. Cell 20, 29–36 (1980).
Reichardt, L. F. & Patterson, P. H. Nature 270, 147–151 (1977).
Gozes, I. & Richter-Landsberg, C. FEBS Lett. 95, 169–172 (1978).
Gozes, I., Saya, D. & Littauer, U. Z. Brain Res. 171, 171–175 (1979).
Gozes, I. & Littauer, U. Z. FEBS Lett. 99, 86–90 (1979).
Zisapel, N., Levi, M. & Gozes, I. J. Neurochem. 34, 26–32 (1980).
Campenot, R. B. Meth. Enzym. 53, 302–307 (1979).
Brady, S. T., Tytell, M., McQuarrie, I. G. & Lasek, R. J. J. Cell Biol. 187, 250a (1980).
Kirschner, M. W. Int. Rev. Cytol. 54, 1–71 (1978).
Marek, A., Fellous, A., Francon, J. & Nunez, J. Nature 284, 353–355 (1980).
Hawrot, E. & Patterson, P. H. Meth. Enzym. 53, 574–584 (1979).
Jacobs, M., Brown, A. V. W. & Brown, G. L. FEBS Lett. 24, 113–116 (1972).
O'Farrell, P. H. J. biol. Chem. 250, 4007–4021 (1975).
Laskey, R. A. & Mills, A. D. FEBS Lett. 82, 314–316 (1977).
Ludnena, R. F. & Woodward, D. O. Proc. natn. Acad. Sci. U.S.A. 70, 3594–3598 (1973).
Lu, R. C. & Elzina, M. Biochim. biophys. Acta 537, 320–328 (1978).
Valenzuela, P. et al. Nature 289, 650–655 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gozes, I., Sweadner, K. Multiple tubulin forms are expressed by a single neurone. Nature 294, 477–480 (1981). https://doi.org/10.1038/294477a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/294477a0
This article is cited by
-
Deciphering the Enigma: NAP (CP201) the Active ADNP Drug Candidate Enters Cells by Dynamin-Associated Endocytosis
Journal of Molecular Neuroscience (2020)
-
Microtubule Destabilization Paves the Way to Parkinson’s Disease
Molecular Neurobiology (2017)
-
Sexual divergence in microtubule function: the novel intranasal microtubule targeting SKIP normalizes axonal transport and enhances memory
Molecular Psychiatry (2016)
-
Tau pathology: predictive diagnostics, targeted preventive and personalized medicine and application of advanced research in medical practice
EPMA Journal (2010)
-
NAP protects against cyanide-related microtubule destruction
Journal of Neural Transmission (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.