Abstract
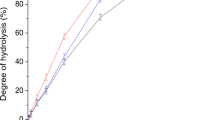
DURING an investigation of the early stages of hydrolysis of various mucoids and connective-tissue proteins, it was observed that, on partial hydrolysis with acetic acid or oxalic acid at 100°, aspartic acid was in each case the first amino-acid to appear in the free condition, followed at a somewhat later stage by glutamic acid1. Since elastin has an unusually low content of the dicarboxylic amino-acids2, it was thought that, with this protein, hydrolytic cleavage at some proportion of the aspartic or glutamic acid residues might yield soluble degradation products of high molecular weight.
Similar content being viewed by others
Article PDF
References
Partridge, S. M., and Davis, H. F., Nature, 165, 62 (1950).
Neuman, R. E., Arch. Biochem., 24, 289 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ADAIR, G., DAVIS, H. & PARTRIDGE, S. A Soluble Protein derived from Elastin. Nature 167, 605 (1951). https://doi.org/10.1038/167605a0
Issue Date:
DOI: https://doi.org/10.1038/167605a0
This article is cited by
-
Fluid protein condensates for bio-inspired applications
Nature Reviews Bioengineering (2023)
-
Histochemie des Bindegewebes
Archiv f�r Klinische und Experimentelle Dermatologie (1957)
-
Structure of Elastic Tissue
Nature (1952)
-
Free Amino Groups of Collagen
Nature (1951)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.