Summary
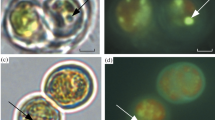
Vegetative cells and heterocysts of the filamentous desiccation-tolerant cyanobacteriumNostoc commune HUN retain their ultrastructural organisation and the integrity of their intra- and extracellular membranes after two years of desiccation and subsequent rehydration. Immunogold-labelling of thin sections demonstrated the presence of NifH (Fe protein of nitrogenase) in vegetative cells and heterocysts within five minutes of the rehydration of dried colonies. Immunogold label accumulated in discrete areas vegetative cells within 5 minutes of the rewetting of cells, and after 30 minutes a conspicuous association of NifH protein with heterocyst ribosomes was detected. After longer periods of rehydration, the deposition of gold particles became more random within both cell types but occurred with a greater frequency in heterocysts. Up to 24 hours after the rewetting of cells, two morphologically-distinct forms of heterocyst could be discerned. NifH protein was detected through Western blotting (subunit Mr=33,800) in protein extracts from samples ofNostoc commune, collected in different parts of the world and including some which had been desiccated for periods of up to 10 years. The results are discussed in relation to the sequential recovery of metabolic functions, particularly nitrogen fixation, which occurs upon the rehydration of cells after their prolonged storage in the air-dry state.
Similar content being viewed by others
References
Angeloni SV, Potts M (1987) Polysome turnover in immobilized cells ofNostoc commune (Cyanobacteria) exposed to water stress. J Bacteriol 168: 1036–1039
Bewley JD (1979) Physiological aspects of desiccation tolerance. Ann Rev Plant Physiol 30: 195–238
Carr NG, Whitton BA (1982) The biology of cyanobacteria. Blackwell Scientific Publications, Oxford London Edinburgh Boston Melbourne, pp 688
Defrancesco N, Potts M (1988) Cloning ofnifHD fromNostoc commune UTEX 584 and a flanking region homologous to part of theAzotobacter vinelandii nifU gene. J Bacteriol 170: 3297–3300
Ferrari-Iliou R, Pham Thi AT, Dasilva V (1984) Effect of water stress on the lipid and fatty acid composition of cotton (Gos-sypium hirsutum) chloroplasts. Physiol Plant 62: 219–224
Helber JT, Johnson TR, Yarbrough LR, Hirschberg R (1988) Regulation of nitrogenase gene expression in anaerobic cultures ofAnabaena variabilis. J Bacteriol 170: 552–557
Kallas T, Rippka R, Coursin T, Rebiere M-C, Tandeaude Marsac N, Cohen Bazire G (1983) Aerobic nitrogen fixation by non-heterocystous cyanobacteria. In:Papageorgiou GC, Packer L (eds) Photosynthetic prokaryotes: cell differentiation and function. Elsevier, New York, pp 281–303
Leopold AC (1986) Membranes, metabolism and dry organisms. Cornstock Publishing Associates (Branch of Cornell University Press), pp 374
Lynn ME, Ownby JD (1987) Transcriptional activity of heterocysts isolated fromAnabaena variabilis. Arch Microbiol 148: 115–120
Peat A, Potts M (1987) The ultrastructure of immobilised desiccated cells of the cyanobacteriumNostoc commune UTEX 584. FEMS Microbiol Lett 43: 223–227
Potts M, Bowman MA (1985) Sensitivity ofNostoc commune UTEX 584 (Cyanobacteria) to water stress. Arch Microbiol 141: 51–56
— (1985) Protein synthesis and proteolysis in immobilized cells of the cyanobacteriumNostoc commune UTEX 584 exposed to matrix water stress. J Bacteriol 164: 1025–1031
— (1986) The protein index ofNostoc commune UTEX 584 (Cyanobacteria): changes induced in immobilised cells by water stress. Arch Microbiol 146: 87–95
—,Olie JJ, Nickels JS, Parsons J, White DC (1987) Variation in phospholipid ester-linked fatty acids and carotenoids of desiccatedNostoc commune (Cyanobacteria) from different geographic locations. Appl Environ Microbiol 53: 4–9
Rippka R, Stanier RY (1978) The effects of anaerobiosis on nitrogenase synthesis and heterocyst development by Nostocacean Cyanobacteria. J Gen Microbiol 105: 83–94
Scherer S, Ernst A, Chen T-W, Böger P (1984) Rewetting of drought-resistant blue-green algae: Time course of water uptake and reappearance of respiration, photosynthesis, and nitrogen fixation. Oecologia (Berl) 62: 418–423
Smith RL, van Baalen C, Tabita FR (1987) Alteration of the Fe protein of nitrogenase by oxygen in the cyanobacteriumAnabaena sp. strain CA. J Bacteriol 169: 2537–2542
Stal LJ, Krumbein WE (1985) Oxygen protection of nitrogenase in the aerobicaly nitrogen fixing, non-heterocystous cyanobacteriumOscillatoria sp. Arch Microbiol 143: 72–76
Symington J (1984) Electrophoretic transfer of proteins from two-dimensional gels to sheets and their detection. In:Celis JE, Bravo R (eds) Two -dimensional gel electrophoresis of proteins. Academic Press, New York, pp 127–168
Thompson SW (1969) Selected histochemical and histopathological methods. CC Thomas, Springfield
Titus DE, Becker WM (1985) Investigation of the glyocysome-peroxisome transition in germinating cucumber cotyledons using double-label immunoelectron microscopy. J Cell Biol 101: 288–1299
Weisshaar H, Boger P (1983) Nitrogenase activity of the non-heterocystous cyanobacteriumPhormidium foveolarum. Arch Microbiol 136: 270–274
Whitton BA, Donaldson AD, Potts M (1979) Nitrogen fixation byNostoc colonies in terrestial environments of Aldabra Atoll, Indian Ocean. Phycologia 18: 278–287
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peat, A., Powell, N. & Potts, M. Ultrastructural analysis of the rehydration of desiccatedNostoc commune HUN (cyanobacteria) with particular reference to the immunolabelling of NifH. Protoplasma 146, 72–80 (1988). https://doi.org/10.1007/BF01405915
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01405915