Abstract
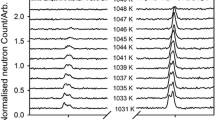
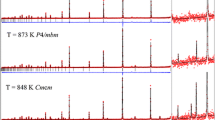
The crystal structures of members of the åkermanite-gehlenite solid solution series have been refined from high resolution neutron powder diffraction data. Synthetic samples of 0, 25, 50, 75 and 100% åkermanite composition were used. The results are consistent with the ordering scheme for the tetrahedrally-coordinated cations represented by the structural formula Ca2[Mg x Al1-x]1[Si1+x Al1-x]2O7 where the superscripts denote inequivalent tetrahedral sites. No evidence was found for the existence of phase transitions in the samples with x ≤ 0.75, in contrast to the behaviour found in pure åkermanite. The relationship between the cell parameters of meilite and the tetrahedral conformations and the resulting non-ideality of the solid-solution are discussed.
Similar content being viewed by others
References
Armbruster T, Roethlisberger F, Seifert S (1990) Layer topology, stacking variation and site distortion in melilite-related compounds in the system CaO-ZnO-GeO2-SiO2. Am Mineral 75:847–858
Brown ID (1981) The Bond-Valence Method: An Empirical Approach to Chemical Structure and Bonding Structure. Chapter14. In: O'Keefe M, Navrotsky A (eds) Structures and Bonding in Crystals II, Academic Press, New York, pp 1–30
Brown ID, Altermatt D (1985) Bond-valence Parameters Obtained from a Systematic Analysis of the Inorganic Crystal Structure Database. Acta Crystallogr B41:244–247
Charlu TV, Newton RC, Kleppa OJ (1981) Thermochemistry of synthetic Ca2Al2SiO7 (Gehlenite)-Ca2MgSi2O7 (Åkermanite) Melilites. Geochim Cosmochim Acta 45:1609–1617
David WIF, Akporiaye DE, Ibberson RM, Wilson CC (1988) The High Resolution Powder Diffractometer at ISIS — An Introductory Users Guide. Version 1.0. Rutherford Appleton Laboratories, Chilton, Didcot, Oxon
Deer WA, Howie RA, Zussman J (1986) Rock Forming Minerals.Volume 1 B Disilicates and Ring Silicates. Longman, UK
Hemingway BS, Evans HT Jr, Nord GL Jr, Haselton HT Jr, Robie RA, McGee JJ (1986) Åkermanite: Phase transitions in heat capacity and thermal expansions, and revised thermodynamic data. Can Mineral 24:425–434
Kimata M, Ii N (1981) The crystal structure of synthetic Åkermanite Ca2MgSi2O7. N Jahrb Mineral Mh 1:1–10
Kimata M, Ii N (1982) The structural properties of synthetic Gehlenite Ca2Al2SiO7. N Jahrb Mineral Abh 144:254–267
Lachowski EE, Glasser FP (1973) Applications of gas chromatography to mineral chemistry: Aluminium-Silicon ordering in melilites. Mineral Mag 39:412–419
Raaz F (1930) Die Struktur des synthetischen Gehlenit, 2CaO.Al2O3.SiO2 Anz Akad Wiss Wien, Math-Naturwiss Kl 18:203–211
Schmahl WW (1990) Search Tree Method for the Determination of Similarity Operators between Arbitrary Lattices. Z Kristallogr 191:23–38
Seifert F, Czank M, Simons B, Schmahl W (1987) A Commensurate-Incommensurate Phase Transition in Iron-Bearing Åkermanites. Phys Chem Minerals 14:26–35
Smith JV (1953) Re-examination of the crystal structure of melilite. Am Mineral 38:643–661
Waldbaum DR, Woodhead JA (1975) Thermodynamic and crystallographic properties related to Al-Si-Mg ordering in Ca2MgSi2O7.-Ca2Al2SiO7 melilites crystalline solutions. Fortschr Miner 52:119–131
Warren BE (1930) The structure of melilite (Ca, Na)2(Mg, Al)(Si, Al)2O7. Z Kristallogr 74:131–138
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Swainson, I.P., Dove, M.T., Schmahl, W.W. et al. Neutron powder diffraction study of the åkermanite-gehlenite solid solution series. Phys Chem Minerals 19, 185–195 (1992). https://doi.org/10.1007/BF00202107
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202107