Summary
The single-dose plasma kinetics of diflunisal was studied in healthy young and old subjects, in patients with rheumatoid arthritis, and in patients with renal failure. The plasma and urine kinetics of the glucuronidated metabolites of diflunisal were studied in the healthy elderly subjects and in the patients with renal failure. In addition, the multiple-dose plasma kinetics of diflunisal was assessed in healthy volunteers and in patients with rheumatoid arthritis.
After a single dose of diflunisal the terminal plasma half-life, mean residence time and apparent volume of distribution were higher in elderly subjects than in young adults. No difference was observed in any pharmacokinetic parameter between age-matched healthy subjects and patients with rheumatoid arthritis.
The elimination half-life of unchanged diflunisal was correlated with the creatinine clearance (r=+0.89) and its apparent total body clearance exhibited linear dependence on creatinine clearance (r=+0.78).
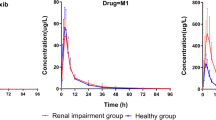
In patients with renal failure, the terminal plasma half-life and mean residence time of diflunisal were prolonged. The renal and apparent total body clearances were lower, the mean apparent volume of distribution was higher and the mean area under the concentration-time curve extrapolated to infinity (AUC) was greater in the renal failure patients than in controls.
The plasma concentration of the glucuronidated metabolites rapidly rose to levels above those of unchanged drug in renal patients, whereas they were lower than those of unchanged diflunisal in controls. The AUC (0–96 h) of diflunisal glucuronides in the patients was four-times that in controls, and the terminal elimination half-life of the glucuronides was prolonged in them. The renal excretion and clearance of diflunisal glucuronides were reduced when renal function was impaired.
After multiple dosing, the pre-dose steady-state plasma-concentration increased with decreasing creatinine clearance (r=-0.79). When the plasma concentration exceeded 200 µmol·1−1, the elimination half-life was doubled, due to partial saturation of diflunisal conjugation. This finding suggests that lower doses could be used in long-term treatment.
Thus, old age and arthritic disease appear to have little influence on the kinetics of diflunisal in the absence of renal functional impairment. Ordinary doses can be given for short term treatment of elderly patients with or without RA. In patients with renal failure, however, reduced doses of diflunisal are recommended.
Similar content being viewed by others
References
Brune K, Graf P, Rainsford KD (1977) A pharmacokinetic approach of the understanding of therapeutic effects and side-effects of salicylates. In: Rainsford KD, Brune K, Whitehouse MW (eds) Aspirin and related drugs: Their actions and uses. Agents Actions [Suppl] 1: 9–26
Brune K, Rainsford KD, Schweitzer A (1980) Biodistribution of mild analgesics. Br J Clin Pharmacol 10: 2795–2845
Umbenhauer ER (1983) Diflunisal in the treatment of pain of osteoarthritis. Pharmacother 3: 555–605
Besselar G, Steelman SL, Tempero KF, Honig W, Brosens Y, Candron J, De Vroey P (1975) Analgesic efficacy of MK-647, a novel salicylate. Clin Pharmacol Ther 17: 229
Steelman SL, Tempers KF, Besselaar GH, Honig W, De Vroey P, Bahons I, Muller W, Cheries A (1975) The analgesic activity of diflunisal, a novel salicylate, in various pain models. Clin Res 253: 224
Patrono C, Ciabattoni G, Patrignani P (1985) Clinical pharmacology of platelet cyclooxygenase inhibition. Circulation 72: 1177–1184
Smith Sibinga CT (1977) Effect of diflunisal on platelet function and blood coagulation. Br J Clin Pharmacol 4: 375–385
Green DL, Davies RO, Holmes GI, Kohl H, Lee RB, Reynolds N, Schmid FR, Tsao C (1981) Effect of diflunisal on platelet function and fecal blood loss. Clin Pharmacol Ther 30: 378–384
Dieppe PA (1978) Nephrotoxicity studies on aspirin and diflunisal. Curr Med Res Opin 5: 575–579
Dieppe PA, Doyle DV, Burry HC (1979) Renal damage during tresatment with antirheumatic drugs. Br Med J 2: 664
Dieppe PA, Huskisson EC (1979) Diflunisal and aspirin: A comparison of efficacy and nephrotoxicity in osteoarthritis. Rheumatol Rehabil 18: 53–56
Lindholm L, Ekdahl C, Håkansson E, Melander A, Olsson S, Svensson B, Wåhlin-Boll E (1981) Comparison of diflunisal and acetylsalicylic acid in patients with rheumatoid arthritis. Scand J Rheumatol 10: 289–295
De Silva M, Hazleman BL, Dippy JE (1980) Diflunisal and aspirin: A comparative study in rheumatoid arthritis. Rheumatol Rehabil 19: 126–130
Caruso I, Bianchi-Porro G (1980) Gastroscopic evaluation of anti-inflammatory agents. Br Med J 280: 75–78
Cockel R, Kendall MJ, Becker JF, Hawkins CF (1971) Serum biochemical values in rheumatoid disease. Ann Rheum Dis 30: 166–170
Schneider RE, Bishop H, Kendall MJ, Quarterman CP (1981) Effect of inflammatory disease on plasma concentrations of three beta-adrenoreceptor blocking drugs. Int J Clin Pharmacol Ther Toxicol 19: 158–162
Crock PR, Willis JV, Kendall MJ, Jack DB, Fowler PD (1982) The pharmacokinetics of diclofenac sodium in patients with active rheumatoid disease. Eur J Clin Pharmacol 21: 331–334
Rainford KD, Schweitzer A, Brune K (1981) Autoradiographic and biochemical observations on the distribution of nonsteroid antiinflammatory drugs. Arch Int Pharmacodyn 250: 180–194
Tocco DJ, Breault GO, Zacchei AG, Steelman SL, Derrier CV (1975) Physiological disposition and metabolism of 5-(2′,4′-difluoraphenyl) salisylic acid, a new salicylate. Drug Metab Dispos 3: 453–456
Yeh KE, Tocco DJ (1982) Nonlinear pharmacokinetics of diflunisal (abstr.) Proc Acad Pharm Sci 52
Lin JH, Hook KF, Yea KC, Duggan DE (1985) Dose-dependent pharmacokinetics of diflunisal in rats: Dual effects of protein binding and metabolism. J Pharmacol Exp Ther 235: 402–406
Verbeeck R, Tjandramaga TB, Mullie A, Verbesselt R, Verberckmoes R, De Schepper PJ (1979) Biotransformation of diflunisal and renal excretion of its glucoronides in renal insufficiency. Br J Clin Pharmacol 7: 273–282
Elseviers MM, Verpooten GA, De Broe ME (1987) Interpretation of creatinine clearance. Lancet: 457
Wåhlin-Boll E, Brantmark B, Hanson A, Melander A, Nilsson C (1981) High-pressure liquid chromatographic determination of acetylsalicylic acid, salicylic acid, diflunisal, indomethacin, indoprofen and indobufen. Eur J Clin Pharmacol 20: 375–378
Verbeeck R, Tjandramaga TB, Mullie A, Verbesselt R, De Schepper PJ (1979) Effect of aluminium hydroxide on diflunisal absorption. Br J Clin Pharmacol 7: 519–522
Tobert JA, De Schepper PJ, Tjandramaga TB, Mullie A, Buntinx AP, Meisinger MAP, Huber PB, Hall TLP, Yeh KC (1981) Effect of antacids on the bioavailability of diflunisal in the fasting and postprandial states. Clin Pharmacol Ther 30: 385–389
Dettli L (1974) Individualization of drug dosage in patients with renal disease. Med Clin North Am 58: 977–985
Spring P (1975) Calculation of drug dosage regimens in patients with renal disease: A new nomographic method. Int J Clin Pharmacol 11: 76–80
Bayles TB (1962) Plasma salicylate levels in rheumatoid arthritis. In: Dixon A, Martin BK, Smith MJH, Wood PHN (eds) Salicylates, an international symposium. Churchill, London, pp 43–46
Grindel JM, Migdalof BH, Plostnieks J (1979) Absorption and excretion of tolmetin arthritic patients. Clin Pharmacol Ther 26: 122–128
Selley ML, Glass J, Triggs EJ, Thomas J (1975) Pharmacokinetic studies of tolmetin in man. Clin Pharmacol Ther 17: 599–605
Chalmers IM, Pohl JEF, Platt DS (1969) The evaluation in man of fenclozic acid, a new anti-inflammatory agent. Serum concentration studies in healthy individuals and in patients with rheumatoid arthritis. Ann Rheum Dis 28: 590–594
Alván G, Orme M, Bertilsson L, Ekstrand R, Palmér L (1975) Pharmacokinetics of indomethacin. Clin Pharmacol Ther 18: 364–373
Baber N, Halliday LDC, Van Den Heuvel WJA, Walker RW, Sibeon R, Keenan HP, Littler T, Orme M (1979) Indomethacin in rheumatoid arthritis: Clinical effects, pharmacokinetics and non-responders. Ann Rheum Dis 38: 128–137
Brodie BB, Lowman EW, Burns JJ, Lee PR, Chenhin T, Goldman A, Weiner M, Steele MJ (1954) Observations on the anti-rheumatic and physiologic effects of phenylbutazone and some comparisons with cortisone. Am J Med 16: 181–190
Aarbacke J, Bakke OM, Milde EJ, Davies DS (1977) Disposition and oxidative metabolism of phenylbutazone in man. Eur J Clin Pharmacol 11: 359–366
Rooney L, Kendall MJ, Main A, Hosie J, John VA (1985) Pharmacokinetics of pirprofen in young volunteers and elderly patients. Eur J Clin Pharmacol 29: 73–77
Verbeeck RK, De Schepper PJ (1980) Influence of chronic renal failure och hemodialysis on diflunisal plasma protein binding. Clin Pharmacol Ther 27: 628–635
Dettli L (1977) Elimination kinetics and dosage adjustment of drugs in patients with kidney disease. Progr Pharmacol 1: 8–17
Shimomura K, Kamata O, Keki S, Ida S, Oguri K, Yoshimura H, Tsukamoto H (1971) Analgesic effect of morphine glucuronides. Tohoku J Exp Med 105: 45–52
Osborne RJ, Joel SP, Slevin ML (1986) Morphine intoxication in renal failure: The role of morphine-6-glucuronide. Br Med J 292: 1548–1549
Levy G (1979) Decreased body clearance of diflunisal in renal insufficiency — an alternative explanation. Br J Clin Pharmacol 8: 601
Faed EM (1980) Decreased clearance of diflunisal in renal insufficiency — an alternative explanation. Br J Clin Pharmacol 10: 185–185
Odar-Cederlöf I, Vessman J, Alvan G, Sjöqvist F (1977) Oxazepam disposition in uremic patients. Acta Pharmacol Toxicol 40: 52–62
Blum M, Bauminger S, Algueti A, Kish E, Ayalon D, Aviram A (1981) Urinary prostaglandin E2 in chronic renal disease. Clin Nephrol 15: 87–89
Garella S, Matarese RA (1984) Renal effects of prostaglandins and clinical adverse effects of non-steroidal anti-inflammatory agents. Medicine 63: 165–181
Carmichael J, Shankel SW (1985) Effects of nonsteroidal anti-inflammatory drugs on prostaglandins and renal function. Am J Med 78: 992–1000
Brune K (1981) Inhibition by anti-inflammatory drugs of prostaglandin production in cultured macrophages. Naunyn Schmiedebergs Arch Pharmacol 315: 269–279
Majerus PW, Stanford N (1977) Comparative effects of aspirin and diflunisal on prostaglandin synthetase from human platelets and sheep seminal vesicles. Br J Clin Pharmacol 4: 155–183
Shen TY, Ham EA, Cirillo VJ, Zanetti M (1974) Structure-activity relationship of certain prostaglandin synthetase inhibitors. In: Robinson HJ, Vane IR (eds) Prostaglandin synthetase inhibitors. Raven, New York, pp 19–31
Shen TY (1977) Prostaglandin synthetase inhibitors. In: Berti F, Samuelsson B, Velo GP (eds) Prostaglandin and tromboxanes. NATO advanced study institute series. A life sciences. Plenum, New York, pp 111–136
Schultz P, Perrier CV, Ferber-Perret F, Van den Heuvel WJ, Steelman SL (1979) Diflunisal, a new acting analgesic and prostaglandin inhibitor: Effect of concomitant acetylsalicylic acid therapy on ototoxicity and on disposition of both drugs. J Int Med Res 7: 61–68
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eriksson, L.O., Wåhlin-Boll, E., Odar-Cederlöf, I. et al. Influence of renal failure, rheumatoid arthritis and old age on the pharmacokinetics of diflunisal. Eur J Clin Pharmacol 36, 165–174 (1989). https://doi.org/10.1007/BF00609190
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609190