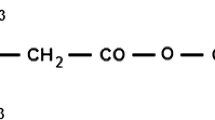Abstract
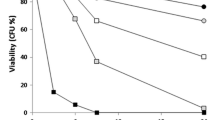
Haploid cells of opposite mating type of Saccharomyces cerevisiae conjugate to form zygote. During the conjugation process, the degradation or reorganization of the cell wall and the fusion of the two plasma membranes take place. Since chloroquine inhibits cellular events associated with the reorganization of the plasma membrane, the effect of the drug on conjugation was studied. Chloroquine at a concentration, at which cell growth was not retarded, inhibited zygote formation, while it did not affect other mating functions, such as sexual agglutination, production of and response to mating pheromone. Cells in a mating culture containing chloroquine formed no “prezygote” suggesting that they were not prepared for entering into fusion process. The inhibitory effect of chloroquine was reversible as cells formed zygote when they were washed after treatment with chloroquine. Zygote formation was unaffected in cells possessing chlorquine within vacuoles after incubation with the drug in complete medium (YPD) at pH 7.5, followed by washing. This suggests that chloroquine inhibits zygote formaton by adsorbing to the plasma membrane of S. cerevisiae.
Similar content being viewed by others
Abbreviations
- YPD:
-
yeast extract-pepton-dextrose medium
References
Ascoli M, Pruett D (1978) Inhibition of the degradation of receptor bound human choriogonadotropin by lysosomotropic agents, protease inhibitors and metabolic inhibitors. J Biol Chem 253:7832–7838
Ciechanover AJ, Schwartz AL, Dautry-Varsat A, Lodish HF (1983) Kinetics of internalization and recycling of the transferrin receptor in a human hepatoma cell line: effect of lysosomotropic agents. J Biol Chem 258:9681–9689
De Duve C, De Barsy T, Poole B, Trouet A, Tulkens P, Van Hoof F (1974) Lysosomotropic agents. Biochem Pharmacol 24:2495–2531
Di Donato S, Wiesmann UN, Herschkowitz N (1977) Membrane adsorption and internalization of [14C] chloroquine by cultured human fibroblasts. Biochem Pharmacol 26:7–10
Doi S, Yoshimura M (1978) Temperature-dependent conversion of sexual agglutinability in Saccharomyces cerevisiae. Mol Gen Genet 162:251–257
Doi S, Suzuki Y, Yoshimura M (1979) Induction of sexual cell agglutinability of a mating type cells by α-factor in Saccharomyces cerevisiae. Biochem Biophys Res Commun 91:849–853
Gonzales-Noriega A, Grubb JH, Talkad V, Sly WS (1980) Chloroquine inhibits lysosomal enzyme pinocytosis and enhances lysosomal enzyme secretion by imoairing receptor recycling. J Cell Biol 85:839–852
Kaplan J, Keogh EA (1981) Analysis of the effects of amines on inhibition of receptor-mediated and fluid-phase pinocytosis in rabbit alveolar macrophages. Cell 24:925–932
Lenz AG, Holzer H (1984) Effects of chloroquine on proteolytic processes and energy metabolism in yeast. Arch Microbiol 137:104–108
Lippincott-Schwartz J, Fambrough DM (1987) Cycling of the integral membrane glycoprotein, LEP 100, between plasma membrane and lysosomes: Kinetic and morphological analysis. Cell 49:669–677
Maxfield FR (1982) Weak bases and ionophores rapidly and reversibly raise the pH endocytic vesicles in cultured mouse fibroblasts. J Cell Biol 95:676–681
Mellman I, Fuchs R, Helenius A (1986) Acidification of the endocytic and exocytic pathways. Annu Rev Biochem 55:663–700
Ohkuma S, Poole B (1978) Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci USA 75:3327–3331
Ohkuma S, Poole B (1981) Cytoplasmic vacuolation of mouse peritoneal macrophages and the uptake into lysosomes of weakyl basic substances. J Cell Biol 90:656–664
Poole B, Ohkuma S (1981) Effect of weak bases on the intralysosomal pH in mouse peritoneal macrophages. J Cell Biol 90:665–669
Sando GN, Titus-Dillon P, Hall CW, Neufeld EF (1979) Inhibition of receptor mediated uptake of a lysosomal enzyme into fibroblasts by chloroquine, protaine and ammonia. Exp Cell Res 119:359–364
Schwartz AL, Bolognesi A, Fridovich SE (1984) Recycling of the asialoglycoprotein receptor and the effect of lysosomotropic amines in hepatoma cells. J Cell Biol 98:732–738
Strous GJ, Du Maine A, Zijderhand-Bleekemolen JE, Slot JW, Schwartz AL (1985) Effect of lysosomotropic amines on the secretory pathway and on the recycling of the asialoglycoprotein receptor in human hepatoma cells. J Cell Biol 101:531–538
Tietze C, Schlesinger P, Stahl P (1980) Chloroquine and ammonium ion inhibit receptor mediated endocytosis of mannose glycoconjugates by macrophages: apparent inhibition of receptor recycling. Biochem Biophys Res Commun 93:1–8
Trueheart J, Boeke JD, Fink GR (1987) Two genes required for cell fusion during yeast conjugation: Evidence for a pheromone-induced surface protein. Mol Cell Biol 7:2316–2328
Wibo M, Poole B (1974) Protein degradation in cultured cells. II. The uptake of chloroquine by rat fibroblasts and the inhibition of cellular protein degradation and cathepsin B1. J Cell Biol 63:430–440
Yanagishima N, Yoshida K, Hamada K, Hagiya M, Kawanabe Y, Sakurai A, Tamura S (1976) Regulation of sexual agglutinability in Saccharomyces cerevisiae of a and α types by sexspecific factors produced by their respective opposite mating types. Plant Cell Physiol 17:439–450
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doi, S., Tanabe, K., Watanabe, M. et al. Chloroquine, a lysosomotropic agent, inhibits zygote formation in yeast. Arch. Microbiol. 151, 20–25 (1988). https://doi.org/10.1007/BF00444663
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00444663