Abstract
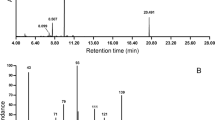
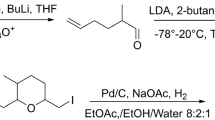
The major sex attractant of the oak leaf roller (Archips semiferanus Walker),cis-10-tetradecenyl acetate (I), was synthesized in 55–62% overall yield by a highly economical route from inexpensive azelaic acid (II).
Similar content being viewed by others
References
Butenandt, A., Hecker, E., Hopp, M., andKoch, W. 1962. Die synthese des bombykols und dercis-trans-isomerm hexadecadien-(10,12)-ole-(l).Justus Liebig's Ann. Chem. 658:39–64.
Chutt, P., andHausser, J. 1929. Sur les acides-alcools polyméthylène-carboniques de 8 à 21 atomes de carbone.Helv. Chim. Acta. 12:463–492.
Cram, D.J., andAllinger, N. 1956. Macro rings. XIII. Synthesis and properties of 1,7-cyclododecadiyne and related compounds.J. Am. Chem. Soc. 78:2518–2524.
Hendry, L.B., Anderson, M.E., Jugovich, J., Mumma, R.O., Robacker, D., andKosarych, Z. 1975. Sex pheromone of the oak leaf roller—A complex chemical messenger system identified by mass fragmentography.Science 187:355–357.
Hendry, L.B., Capello, L., andMumma, R.O. 1974a. Sex attractant trapping techniques for the oak leaf roller,Archips semiferanus Walker.Melsheimer Ser. 16:1–9.
Hendry, L.B., Gill, R.J., Santora, A., andMumma, R.O. 1974b. Sex pheromones in the oak leaf roller(Archips semiferanus): Isolation and field studies.Entomol. Exp. Appl. 17:459–467.
Hendry, L.B., Jugovich, J., Roman, L., Anderson, M.E., andMumma, R.O. 1974c.cis-10-Tetradecenyl acetate, an attractant component in the sex pheromone of the oak leaf roller moth(Archips semiferanus Walker).Experientia 30:886–888.
Hendry, L.B., Roman, L., andMumma, R.O. 1973. Evidence of a sex pheromone in the oak leaf roller,Archips semiferanus (Lepidoptera:Tortricidae): laboratory and field bioassays.Environ. Entomol. 2:1024–1028.
Huber, W.F. 1951. A study of n-octadecenoic acids. I. Synthesis ofcis- andtrans-7-through 12- and of 17-octadecenoic acids.J. Am. Chem. Soc. 73:2730–2733.
Nystrom, R.F., andBrown, W.G., 1947. Reduction of organic compounds by lithium aluminium hydride. I. Aldehydes, ketones, esters, acid chlorides, and acid anhydrides.J. Am. Chem. Soc. 69:1197–1199.
Vogel, A.I. 1934. Physical properties and chemical constitution. Part I. Esters of normal dibasic acids and of substituted malonic acids.J. Chem. Soc. (London) 1934:333–341.
Author information
Authors and Affiliations
Additional information
Authorized as Paper No. 4779 in the Journal Series of the Pennsylvania Agricultural Experiment Station.
Rights and permissions
About this article
Cite this article
Hendry, L.B., Korzeniowski, S.H., Hindenlang, D.M. et al. An economical synthesis of the major sex attractant of the oak leaf roller—cis-10-tetradecenyl acetate. J Chem Ecol 1, 317–322 (1975). https://doi.org/10.1007/BF00988833
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00988833