Abstract
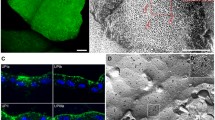
Mammalian urothelium undergoes unique membrane specialization during terminal differentiation making numerous rigid-looking membrane plaques (0.3–0.5 μm diameter) that cover the apical cell surface. The outer leaflet of these membrane plaques is almost twice as thick as the inner leaflet hence the name asymmetric unit membrane (AUM). Ultrastructural studies established that the outer leaflet of AUM is composed of 16 nm particles forming two dimensional crystals, and that each particle forms a ‘twisted ribbon’ structure. We showed recently that highly purified bovine AUMs contain four major integral membrane proteins: uroplakins Ia (27 kD), Ib (28 kD), II (15 kD) and III (47 kD). Studies of the protease sensitivity of the different subdomains of uroplakins and other considerations suggest that UPIa and UPIb have 4 transmembrane domains, while UPII and UPIII have only one transmembrane domain. Chemical Crosslinking studies showed that UPIa and UPIb, which share 39% amino acid sequence, are topologically adjacent to UPII and UPIII, respectively, thus raising the possibility that there exist two biochemically distinct AUM particles, i.e., those containing UPIa/UPII vs. UPIb/UPIII. Bovine urothelial cells grown in the presence of 3T3 feeder cells undergo clonal growth forming stratified colonies capable of synthesizing and processing all known uroplakins. Transgenic mouse studies showed that a 3.6 kb 5′-flanking sequence of mouse uroplakin II gene can drive the expression of bacterial LacZ gene to express in the urothelium. Further studies on the biosynthesis, assembly and targeting of uroplakins will offer unique opportunities for better understanding the structure and function of AUM as well as the biology of mammalian urothelium.
Similar content being viewed by others
References
HicksRM (1965) J. Cell Biol. 26: 25–48
KossLG (1969) Lab. Invest. 21: 154–168
ChlapowskiFJ, BonnevilleMA & StaehelinLA (1972) J. Cell. Biol. 53: 92–104
StaehelinLA, ChlapowskiFJ & BonnevilleMA (1972) J. Cell Biol. 53: 73–91
Bonneville MA & Chlapowski FA (1981) J. Cell Biol. 91: 234a
PorterKR & M. A.Bonneville (1963) An introduction to the fine structure of cells and Tissues. Lea and Febiger Publishers, New York
KossLG (1975) Atlas of Tumor Pathology. 2nd Series, Fasc. II. Armed Forces Institute of Pathology, Washington, DC
BryanGT (1983) In: BryanGT & CohenSM (eds) The Pathology of Bladder Cancer (Vol. I: pp. 1–9) CRC Press, Boca Raton, FL
SandbergAA & BergerCS (1994) J. Urol. 151: 545–560
HicksRM & KettererB (1969) Nature 224: 1304–1305
RobertsonJD & VergaraJ (1980) J. Cell Biol. 86: 514–528
BrissonA & WadeRH (1983) J. Mol. Biol. 166: 21–36
TaylorKA & RobertsonJD (1984) J. Ultrastruct. Res. 87: 23–30
WalzT, HanerM, WuX-R, HennC, EngelA, SunT-T & AebiU (1995) J. Mol. Biol. 248: 887–900
LinJ-H, WuX-R, KreibichG & SunT-T (1994) J. Biol. Chem. 269: 1775–1784
WuX-R & SunT-T (1993) J. Cell Sci. 106: 31–43
WuX-R, LInJ-H, WalzT, HanerM, YuJ, AebiU & SunT-T (1994) J. Biol. Chem. 269: 13716–13724
YuJ, LinJ-H, WuX-R & SunT-T (1994) J. Cell Biol. 125: 171–182
KettererB, HicksRM, ChristodoulidesL & BealeD (1973) Biochim. Biophys. Acta 311: 180–190
StubbsCD, KettererB & HicksRM (1979) Biochim. Biophys. Acta 558: 58–72
VergaraJ, ZambranoF, RobertsonJD & ElrodH (1974) J. Cell Biol. 61: 83–94
WuX-R, ManabeM, YuJ & SunT-T (1990) J. Biol. Chem. 265: 19170–19179
YuJ, ManabeM, WuX-R, XuC, SuryaB & SunT-T (1990) J. Cell Biol. 111: 1207–1216
DongJ-T, LambPW, Rinker-SchaefferCW, VukanovicJ, IchikawaT, IsaacsJT & BarrettJC (1995) Science 268: 884–886
RoachPJ (1991) J. Biol. Chem. 266: 14139–14142
CaruthersJS & BonnevilleMA (1977) J. Cell Biol. 73: 382–399
RheinwaldJG & GreenH (1975) Cell 6: 331–343
RheinwaldJG & GreenH (1980) Cell 6: 331–343
SunT-T & GreenH (1976) Cell 9: 511–521
DoranTI, VidrichA & SunT-T (1980) Cell 22: 17–25
SchermerA, GalvinS & SunT-T (1986) J. Cell Biol. 103: 49–62
WeiZ-G, WuR-L, LavkerRM & SunT-T (1993) Invest. Opthalmol. Vis. Sci. 34: 1814–1828
SuryaB, YuJ, ManabeM & SunT-T (1990) J. Cell Sci. 97: 419–432
YuJ, ManabeM & SunT-T (1992) Epithelial Cell Biol. 1: 4–12
SunT-T, EichnerR, CooperD, SchermerA, NelsonWG & WeissRA (1984) In: LevineA, ToppW, VandeWoudeG and WatsonJD (Ed) The Cancer Cell: The Transformed Phenotype (Vol. I, pp. 169–176) Cold Spring Harbor Press, Cold Spring Harbor, NY
SunT-T, TsengSCG, HuangAJ-W, CooperD, SchermerA, LynchMH, WeissR & EichnerR (1985) In: WangE, FischmanD, LiemR & SunT-T (eds) Intermediate Filaments (pp. 307–309) New York Academy of Sciences, New York
GalvinS, LoomisC, ManabeM, DhouaillyD & SunT-T (1988) Adv. Dermatol. 4: 277–299
O'GuinWM, SchermerA, LynchM & SunT-T (1990) In: SteinertPM & GoldmanR (eds) Cellular and Molecular Biology of Intermediate Filaments (pp. 301–334) Plenum, New York
EichnerR, SunT-T & AebiU (1986) J. Cell Biol. 102: 1767–1777
SchermerA, JesterJV, HardyC, MilanoD & SunT-T (1989) Differentiation 42: 103–110
LinJ-H, ZhaoH & SunT-T (1995) Proc. Natl. Acad. Sci. USA 92: 679–683
WuX-R, MedinaJJ & SunT-T (1995) J. Biol. Chem. 270: 29752–29759
AlroyJ & WeinsteinRS (1980) Anat. Rec. 197: 75–83
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sun, TT., Zhao, H., Provet, J. et al. Formation of asymmetric unit membrane during urothelial differentiation. Molecular Biology Reports 23, 3–11 (1996). https://doi.org/10.1007/BF00357068
Issue Date:
DOI: https://doi.org/10.1007/BF00357068