Abstract
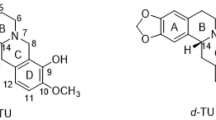
Molecular mechanics calculations using MM3-92 and ab initio quantum mechanical calculations using SPARTAN 5.0 were performed on the structurally similar PCP and BTCP, in which only the latter has a cocaine-like pharmacological profile as a dopamine reuptake blocker. Calculations were also performed on BTCP analogs with a methyl group in various positions of the cyclohexane ring. The results for the cis-2-methyl compound, which retains good pharmacological activity, allowed us to determine that an aryl-axial conformer is the biologically active form for at least some of the compounds in this series. However, an aryl-equatorial conformer presents the identical pharmacophore, as shown by superposition of the two conformers. X-ray crystallographic structures were also obtained for BTCP and related compounds with a 2-methyl group on the cyclohexane ring, with reasonable agreement between the computational and experimental results. Superposition studies were performed with two rigid analogs of cocaine which illustrate the optimal orientations of the ammonium hydrogen for monoamine transporters. There is excellent agreement between a `back-bridged' cocaine analog that is optimal as a dopamine reuptake blocker and the previously proposed biologically active conformer of methylphenidate. However, BTCP is found to be a better fit to the `front-bridged' cocaine analog that is optimal for a serotonin reuptake blocker.
Similar content being viewed by others
References
Johnson, K.M. and Jones, S.M., Annu. Rev. Pharmacol. Toxicol., 30 (1990) 707.
Vincent, J.P., Kartalovski, B., Geneste, P., Kamenka, J.M. and Lazdunski, M., Proc. Natl. Acad. Sci. USA, 76 (1979) 4678.
Vignon, J., Chicheportiche, R., Chicheportiche, M., Kamenka, J.M., Geneste, P. and Lazdunski, M., Brain Res., 280 (1983) 194.
Vignon, J., Pinet, V., Cerruti, C., Kamenka, J.-M. and Chicheportiche, R., Eur. J. Pharmacol., 148 (1988) 427.
He, X.-S., Raymon, L.P., Mattson, M.V., Eldefrawi, M.E. and de Costa, B.R., J. Med. Chem., 36 (1993) 1188.
Ilagouma, M.T., Duterte-Boucher, D., Coderc, E., Vignon, J., Costentin, J. and Kamenka, J.M., Eur. J. Med. Chem., 28 (1993) 377.
Chaudieu, I., Vignon, J., Chicheportiche, M., Kamenka, J.-M., Trouiller, G. and Chicheportiche, R., Pharmacol. Biochem. Behav., 32 (1989) 699.
Froimowitz, M., J. Comput. Chem., 14 (1993) 934.
Froimowitz, M., Patrick, K.S. and Cody, V., Pharm. Res., 12 (1995) 1430.
Froimowitz, M., Deutsch, H.M., Shi, Q., Wu, K.-M., Glaser, R., Adin, I., George, C. and Schweri, M.M., Bioorg. Med. Chem. Lett., 7 (1997) 1213.
Glaser, R., Adin, I., Shiftan, D., Shi, Q., Deutsch, H.M., George, C., Wu, K.-M. and Froimowitz, M., J. Org. Chem., 63 (1998) 1785.
Froimowitz, M. and George, C., J. Chem. Inf. Comput. Sci., 38 (1998) 506.
Musso, D.L., Mehta, N.B., Soroko, F.E., Ferris, R.M., Hollingworth, E.B. and Kenney, B., Chirality, 5 (1993) 495.
Smith, M.P., George, C. and Kozikowski, A.P., Tetrahedron Lett., 39 (1998) 197.
Smith, M.P., Johnson, K.M., Zhang, M., Flippen-Anderson, J.L. and Kozikowski, A.P., J. Am. Chem. Soc., 120 (1998) 9072.
Abraham, P., Pitner, J.B., Lewin, A.H., Boja, J.W., Kuhar, M.J. and Carroll, F.I., J. Med. Chem., 35 (1992) 141.
Kozikowski, A.P., Saiah, M.K.E., Bergmann, J.S. and Johnson, K.M., J. Med. Chem., 37 (1994) 3440.
Madras, B.K., Pristupa, Z.B., Niznik, H.B., Liang, A.Y., Blundell, P., Gonzalez, M.D. and Meltzer, P.C., Synapse, 24 (1996) 340.
Meltzer, P.C., Liang, A.Y., Blundell, P., Gonzalez, M.D., Chen, Z., George, C. and Madras, B.K., J. Med. Chem., 40 (1997) 2661.
Meltzer, P.C., Blundell, P. and Madras, B.K., Med. Chem. Res., 8 (1998) 12.
Meltzer, P.C., Blundell, P., Chen, Z., Yong, Y.F. and Madras, B.K., Bioorg. Med. Chem. Lett., 9 (1999) 857.
Quantum Chemistry Program Exchange, Department of Chemistry, Indiana University, Bloomington, IN.
Serena Software, Bloomington, IN.
Wavefunction Inc., Irvine, CA.
Sheldrick, G.M., SHELXTL97 Program for the Refinement of Crystal Structures, University of Göttingen, Germany, 1997.
Eaton, T.A., Houk, K.N., Watkins, S.F. and Fronczek, F.R., J. Med. Chem., 26 (1983) 479.
Carroll, F.I., Brine, G.A., Boldt, K.G., Mascarella, S.W., Moreland, C.G., Sumner, S.J., Burgess, J.P. and Stejskal, E.O., In Domino, E.F. and Kamenka, J.-M. (Eds.), Sigma and Phencyclidine-like Compounds as Molecular Probes in Biology, NPP Books, Ann Arbor, MI, 1988, pp. 91–106.
Kamenka, J.-M. and Chicheportiche, R., In Domino, E.F. and Kamenka, J.-M. (Eds.), Sigma and Phencyclidine-like Compounds as Molecular Probes in Biology, NPP Books, Ann Arbor, MI, 1988, pp. 1–10.
Manoharan, M., Eliel, E.L. and Carroll, F.I., Tetrahedron Lett., 24 (1983) 1855.
Zenone, F. and Kamenka, J.M., Eur. J. Med. Chem., 26 (1991) 677.
Geneste, P., Kamenka, J.-M., Ung, S.N., Herrmann, P., Goudal, R. and Trouiller, G., Eur. J. Med. Chem., 14 (1979) 301.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Froimowitz, M., Wu, KM., Rodrigo, J. et al. Conformational preferences of the potent dopamine reuptake blocker BTCP and its analogs and their incorporation into a pharmacophore model. J Comput Aided Mol Des 14, 135–146 (2000). https://doi.org/10.1023/A:1008144707255
Issue Date:
DOI: https://doi.org/10.1023/A:1008144707255