Abstract
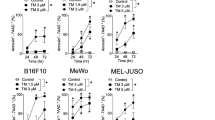
Malignant melanoma is a highly metastatic tumour, resistant to treatment. Serotonin type-3 (5-HT3) receptor antagonists, such as tropisetron and ondansetron, are well-tolerated antiemetic drugs commonly used to prevent nausea caused by chemotherapy or radiotherapy. We investigated the anticancer effects of these drugs on melanoma cancer cell lines WM-266–4 and B16F10 with or without paclitaxel. We constructed IC50 curves and performed Chou–Talalay analysis, using data obtained with the MTT assay. Flow cytometry and fluorescent microscopy were used to examine characteristics of the cell cycle, cell death and cytoskeleton changes. Protein levels and activation were analysed by western blotting and molecular docking studies carried out. Data were analysed by one way ANOVA and post hoc testing. Ondansetron and tropisetron showed selective concentration-dependent cytotoxicity in melanoma cell lines WM-266–4 and B16F10. The effect in combination with paclitaxel was synergistic. The drugs did not cause cell cycle arrest but did promote characteristics of classical apoptosis, including accumulation of subG1 DNA, cleaved caspase-3, mitochondrial membrane permeability and phosphatidylserine exposure. As well, the cytosolic calcium level in the melanoma cells was enhanced, phosphorylated ERK1/2 induced and NF-κB inhibited. Finally, the formation of microtubules was shown to be impaired in melanoma cells treated with ondansetron or tropisetron. Docking studies were used to predict that these drugs could bind to the colchicine binding site on the tubulin molecule. Antiemetic drugs, already given in combination with chemotherapy, may enhance the cytotoxic effect of chemotherapy, following successful delivery to the tumour site.







Similar content being viewed by others
Data availability
The datasets supporting the conclusions of this article are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Abbreviations
- 5-HT3:
-
Serotonin type-3 receptor
- TRO:
-
Tropisetron
- OND:
-
Ondansetron
- NHDF:
-
Normal Human Dermal Fibroblasts
- FBS:
-
Foetal bovine serum
- PTX:
-
Paclitaxel
- PBS:
-
Phosphate-buffered saline
- PFA:
-
Paraformaldehyde
- RT:
-
Room temperature
- PI:
-
Propidium iodide
- TMRE:
-
Tetramethylrhodamine ethyl ester
- BSA:
-
Bovine serum albumin
- SD:
-
Standard deviation
References
Abdel-Aziz H, Windeck T, Ploch M, Verspohl EJ. Mode of action of gingerols and shogoals on 5-HT3 receptors: binding studies, cation uptake by the receptor channel and contraction of isloated guinea-pig ileum. Eur J Pharmacol. 2006;530(1–2):136–43.
Ataee R, Ajdary S, Zarrindast M, Rezayat M, Shokrgozar MA, Ataee A. Y25130 hydrochloride, a selective 5HT3 receptor antagonist has potent antimitogenic and apoptotic effect on HT29 colorectal cancer cell line. Euro J Can Prev. 2010;19:138–43.
Baldwin AS. Control of oncogenesis and cancer therapy resistance by the transcription factor NF-κB. J Clin Invest. 2001;107:241–6.
Bane S. A fluorescence-based high-throughput assay for antimicrotubule drugs. Analytical Biochem. 2003;315:49–56.
Barzegar-Fallah A, Alimoradi H, Mehrzadi S, Barzegar-Fallah N, Zendedel A, Abbasi A, et al. The neuroprotective effect of tropisetron on vincristine-induced neurotoxicity. Neurotoxicol. 2014;41:1–8.
Bower KS, Price KL, Sturdee LEC, Dayrell M, Dougherty DA, Lummis SCR. 5-Fluorotryptamine is a partial agonist at 5-HT3 receptors, and reveals that size and electronegativity at the 5 position of tryptamine are critical for efficient receptor function. Eur J Pharmacol. 2008;580(3):291–7.
Brand MD, Nicholls DG. Assessing mitochondrial dysfunction in cells. Biochem J. 2011;435:297–312.
Chen J, Li CM, Wang J, Ahn S, Wang Z, Lu Y, et al. Synthesis and antiproliferative activity of novel 2-aryl-4-benzoyl-imidazole derivatives targeting tubulin polymerization. Bioorg Med Chem. 2011;19:4782–95.
Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70:440–6.
Cummins DL, Cummins JM, Pantle H, Silverman MA, Leonard AL, Chanmugam A. Cutaneous malignant melanoma. Mayo Clin Proc. 2006;81(4):500–7.
De La Vega L, Muñoz E, Calzado MA, Lieb K, Candelario-Jalil KE, Gschaidmeir H, et al. The 5-HT3 receptor antagonist tropisetron inhibits T cell activation by targeting the calcineurin pathway. Biochem Pharmacol. 2005;70:369–80.
Duan L, Danzer B, Levenson VV, Maki CG. Critical roles for nitric oxide and ERK in the completion of prosurvival autophagy in 4OHTAM-treated estrogen receptor-positive breast cancer cells. Cancer Lett. 2014;353:290–300.
Duan YT, Man RJ, Tang DJ, Yao YF, Tao XX, C. Yu C, , et al. Design, synthesis and antitumor activity of novel link-bridge and B-ring modified combretastatin A-4 (CA-4) analogues as potent antitubulin agents. Sci Rep. 2016;6:25387.
Fadok V, Voelker D, Campbell P, Cohen J, Bratton D, Henson P. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol. 1992;148:2207–16.
Farkas DL, Wei M, Febbroriello P, Carson J, Loew L. Simultaneous imaging of cell and mitochondrial membrane potentials. Biophys J. 1989;56:1053–69.
Gogas HJ, Kirkwood JM, Sondak VK. Chemotherapy for metastatic melanoma: time for a change? Cancer. 2007;109:455–64.
Gray-Schopfer V, Wellbrock C, Marais R. Melanoma biology and new targeted therapy. Nature. 2007;445:851–7.
Grynkiewicz G, Poenie M, Tsien RY. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985;260:3440–50.
Hanwell MD, Curtis DE, Lonie DC, Vandermeersch T, Zurek E, Hutchison GR. Avogadro: an advanced semantic chemical editor, visualization, and analysis platform. J Cheminformatics. 2012;4:17.
Haus U, Späth M, Färber L. Spectrum of use and tolerability of 5-HT3 receptor antagonists. Scand J Rheumatol. 2004;33:12–8.
Hejazi SH, Ahangari G, Deezagi A. Alternative viewpoint against breast cancer based on selective serotonin receptors 5HTR3A and 5HTR2A antagonists that can mediate apoptosis in MCF-7 cell line. Current Drug Discovery Technol. 2015;12:240–9.
Himes RH. Interactions of the catharanthus (Vinca) alkaloids with tubulin and microtubules. Pharmacol Ther. 1991;51:257–67.
Ichas F, Mazat JP. From calcium signaling to cell death: two conformations for the mitochondrial permeability transition pore. Switching from low-to high-conductance state. Biochim Biophys Acta Bioenergetics. 1998;1366:33–50.
Jackman RW, Rhoads MG, Cornwell E, Kandarian SC. Microtubule-mediated NF-kB activation in the TNF-a signling pathway. Exp Cell Res. 2009;315:3242–9.
Jones G, Willett P, Glen RC. Molecular recognition of receptor sites using a genetic algorithm with a description of desolvation. J Mol Biol. 1995;245:43–53.
Jones G, Willett P, Glen RC, Leach AR, Taylor R. Development and validation of a genetic algorithm for flexible docking. J Mol Biol. 1997;267:727–48.
Karin M, Cao Y, Greten FR, Li ZW. NF-κB in cancer: from innocent bystander to major culprit. Nat Rev Can. 2002;2(4):301–10.
Krishan A. Rapid flow cytofluorometric analysis of mammalian cell cycle by propidium iodide staining. J Cell Biol. 1975;66:188–93.
La Regina G, Bai R, Coluccia A, Naccarato V, Famiglini V, Nalli M, et al. New 6-and 7-heterocyclyl-1H-indole derivatives as potent tubulin assembly and cancer cell growth inhibitors. Euro J Med Chem. 2018;152:283–97.
Loew LM, Carrington W, Tuft RA, Fay FS. Physiological cytosolic Ca2+ transients evoke concurrent mitochondrial depolarizations. PNAS. 1994;91:12579–83.
Mebratu Y, Tesfaigzi Y. How ERK1/2 activation controls cell proliferation and cell death: is subcellular localization the answer? Cell Cycle. 2009;8:1168–75.
Molgaard S, Ulrichsen M, Olsen D, Glerup S. Detection of phosphorylated Akt and MAPK in cell culture assays. MethodsX. 2016;3:386–98.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63.
Rello-Varona S, Herrero-Martín D, López-Alemany R, Muñoz-Pinedo C, Tirado OM. “(Not) all (dead) things share the same breath”: identification of cell death mechanisms in anticancer therapy. Cancer Res. 2015;75:913–7.
Roe M, Lemasters J, Herman B. Assessment of Fura-2 for measurements of cytosolic free calcium. Cell Calcium. 1990;11:63–73.
Souphron J, Bodakuntla S, Jijumon AS, Lakisic G, Gautreau AM, Janke C, Magiera MM. Purification of tubulin with controlled post-translational modiciations by polymerization-depolymierization cycles. Nat Prot. 2019;14:1634–60.
Stratz T, Müller W. The use of 5-HT3 receptor antagonists in various rheumatic diseases - a clue to the mechanism of action of these agents in fibromyalgia? Scand J Rheumatol. 2000;29:66–71.
Tang D, Wu D, Hirao A, Lahti JM, Liu L, Mazza B, et al. ERK activation mediates cell cycle arrest and apoptosis after DNA damage independently of p53. J Biol Chem. 2002;277:12710–7.
Tanimura S, Uchiyama A, Watanabe K, Yasunaga M, Inada Y, Kawabata T, Iwashita K, Noda S, Ozaki K, Kohno M. Blockade of constitutively activated ERK signaling enhances cytotoxicity of microtubule-destabilising agents in tumor cells. Biochem Biophys Res Communs. 2009;378:650–5.
Tien S-C, Chang Z-F. Oncogenic Shp2 disturbs microtubule regulation to cause HDAC6-dependent ERK hyperactivation. Oncogene. 2014;33:2938–46.
Vermes I, Haanen C, Steffens-Nakken H, Reutellingsperger C. A novel assay for apoptosis flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled annexin V. J Immunol Methods. 1995;184:39–51.
Wahl AF, Donaldson KL, Fairchild C, Lee FY, Foster SA, Demers GW, Galloway DA. Loss of normal p53 function confers sensitization to Taxol by increasing G2/M arrest and apoptosis. Nat Med. 1996;2(1):72–9.
Wang Y, Zhang H, Gigant B, Yu Y, Wu Y, Chen X, et al. Structures of a diverse set of colchicine binding site inhibitors in complex with tubulin provide a rationale for drug discovery. FEBS J. 2016;283:102–11.
Wolf H. Preclinical and clinical pharmacology of the 5-HT3 receptor antagonists. Scand J Rheumatol. 2000;29:37–45.
Zhao Y, Su J, Goto M, Morris-Natschke SL, Li Y, Zhao QS, Yao ZJ, Lee KH. Dual-functional abeo-taxane derivatives destablising microtubule equilibrium and inhibiting NF-kB activation. J Med Chem. 2013;56:4749–57.
Zhong FJ, Li YM, Xu C, Sun B, Wang JL, Yang LY. EB2 promotes hepatocellular carcinoma proliferation and metastasis via MAPK/ERK pathway by modulating microtubule dynamics. Clin Sci. 2021;135:847–64.
Acknowledgements
The authors would like to thank Dr Joel Tyndall for his kind help with the docking studies. This work was funded by a University of Otago School of Biomedical Sciences’ Dean’s Bequest Grant and a University of Otago Research Grant.
Funding
This work was funded by a University of Otago School of Biomedical Sciences’ Dean’s Bequest Grant and a University of Otago Research Grant.
Author information
Authors and Affiliations
Contributions
ABF and HA designed and carried out the cell culture-based experimental work for this study, analysed the data, and wrote the initial draft of the manuscript. ET carried out the docking studies and SB obtained the funding, was part of designing the experiments, analysing the data, and finalising the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barzegar-fallah, A., Alimoradi, H., Dunlop, J.L. et al. Serotonin type-3 receptor antagonists selectively kill melanoma cells through classical apoptosis, microtubule depolymerisation, ERK activation, and NF-κB downregulation. Cell Biol Toxicol 39, 1119–1135 (2023). https://doi.org/10.1007/s10565-021-09667-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09667-0