Abstract
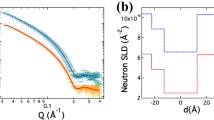
The effect of melatonin and/or cholesterol on the structural properties of a model lipid bilayer prepared from 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) has been investigated both experimentally and via molecular dynamics (MD) simulations. Neutron reflectometry experiments performed with single supported membranes revealed changes in lipid bilayer thickness upon the introduction of additional components. While the presence of cholesterol led to an increase in membrane thickness, the opposite effect was observed in the case of melatonin. The results obtained are in a good agreement with MD simulations which provided further information on the organization of components within the systems examined, indicating a mechanism underlying the membranes’ thickness changes due to cholesterol and melatonin that had been observed experimentally. Cholesterol and melatonin preferentially accumulate in different membrane regions, presumably affecting the conformation of lipid hydrophobic moieties differently, and in turn having distinct impacts on the structure of the entire membrane. Our findings may be relevant for understanding the effects of age-related changes in cholesterol and melatonin concentrations, including those in the brains of individuals with Alzheimer’s disease.






Similar content being viewed by others
References
Avdeev MV, Bodnarchuk VI, Petrenko VI et al (2017) Neutron time-of-flight reflectometer GRAINS with horizontal sample plane at the IBR-2 reactor: possibilities and prospects. Crystallogr Rep 62:1002–1008. https://doi.org/10.1134/S1063774517060025
Belička M, Gerelli Y, Kučerka N, Fragneto G (2015) The component group structure of DPPC bilayers obtained by specular neutron reflectometry. Soft Matter 11:6275–6283. https://doi.org/10.1039/c5sm00274e
Benz RW, Castro-Román F, Tobias DJ, White SH (2005) Experimental validation of molecular dynamics simulations of lipid bilayers: a new approach. Biophys J 88:805–817. https://doi.org/10.1529/biophysj.104.046821
Dies H, Toppozini L, Rheinstädter M (2014) The interaction between amyloid-β peptides and anionic lipid membranes containing cholesterol and melatonin. PLoS ONE 9:e99124. https://doi.org/10.1371/journal.pone.0099124
Drolle E, Kučerka N, Hoopes MI et al (2013) Effect of melatonin and cholesterol on the structure of DOPC and DPPC membranes. Biochim Biophys Acta Biomembr 1828:2247–2254. https://doi.org/10.1016/j.bbamem.2013.05.015
Greenwood AI, Tristram-Nagle S, Nagle JF (2006) Partial molecular volumes of lipids and cholesterol. Chem Phys Lipids 143:1–10. https://doi.org/10.1016/j.chemphyslip.2006.04.002
Hrubovčák P, Kondela T, Ermakova E, Kučerka N (2019) Application of small-angle neutron diffraction to the localization of general anesthetics in model membranes. Eur Biophys J. https://doi.org/10.1007/s00249-019-01370-7
Humphrey W, Dalke A, Schulten K (1996) VMD—{V}isual {M}olecular {D}ynamics. J Mol Graph 14:33–38
Hung W-C, Lee M-T, Chen F-Y, Huang HW (2007) The condensing effect of cholesterol in lipid bilayers. Biophys J 92:3960–3967. https://doi.org/10.1529/biophysj.106.099234
Johnson SJ, Bayerl TM, McDermott DC et al (1991) Structure of an adsorbed dimyristoylphosphatidylcholine bilayer measured with specular reflection of neutrons. Biophys J 59:289–294. https://doi.org/10.1016/S0006-3495(91)82222-6
Kalb E, Frey S, Tamm LK (1992) Formation of supported planar bilayers by fusion of vesicles to supported phospholipid monolayers. Biochim Biophys Acta 1103:307–316. https://doi.org/10.1016/0005-2736(92)90101-q
Kerchner GA, Wyss-Coray T (2016) The role of aging in Alzheimer’s disease BT. In: Sierra F, Kohanski R (eds) Advances in geroscience. Springer, Cham, pp 197–227
Kern W (1990) Evolution of silicon wafer cleaning technology. Proc Electrochem Soc 90:3–19. https://doi.org/10.1149/1.2086825
Klauda JB, Kučerka N, Brooks BR et al (2006) Simulation-based methods for interpreting X-ray data from lipid bilayers. Biophys J 90:2796–2807. https://doi.org/10.1529/biophysj.105.075697
Klauda JB, Venable RM, Freites JA et al (2010) Update of the CHARMM all-atom additive force field for lipids: validation on six lipid types. J Phys Chem B 114:7830–7843. https://doi.org/10.1021/jp101759q
Kreisberg RA, Kasim S (1987) Cholesterol metabolism and aging. Am J Med 82:54–60. https://doi.org/10.1016/0002-9343(87)90272-5
Krueger S, Ankner JF, Satija SK et al (1995) Extending the angular range of neutron reflectivity measurements from planar lipid bilayers: application to a model biological membrane. Langmuir 11:3218–3222. https://doi.org/10.1021/la00008a055
Kučerka N, Nagle JF, Sachs JN et al (2008) Lipid bilayer structure determined by the simultaneous analysis of neutron and X-ray scattering data. Biophys J 95:2356–2367. https://doi.org/10.1529/biophysj.108.132662
Kučerka N, Katsaras J, Nagle JF (2010) Comparing membrane simulations to scattering experiments: introducing the SIMtoEXP software. J Membr Biol 235:43–50. https://doi.org/10.1007/s00232-010-9254-5
Leonard A, Escrive C, Laguerre M et al (2001) Location of Cholesterol in DMPC membranes. A comparative study by neutron diffraaction and molecular mechanics simulation. Langmuir 17:2019–2030
Lindahl E, Abraham MJ, Hess B, Spoel van der D (2020) GROMACS 2020.2 Source code. http://www.gromacs.org/. https://zenodo.org/record/3773801. Accessed 25 Jun 2020
Majkrzak CF, Berk NF (2002) Advances in specular neutron reflectometry. Appl Phys A 74:s67–s69. https://doi.org/10.1007/s003390201876
McLaurin J, Chakrabartty A (1997) Characterization of the interactions of Alzheimer β-amyloid peptides with phospholipid membranes. Eur J Biochem 245:355–363. https://doi.org/10.1111/j.1432-1033.1997.t01-2-00355.x
Murugova T, Ivankov O, Ermakova E et al (2020) Structural changes introduced by cholesterol and melatonin to the model membranes mimicking preclinical conformational diseases. Gen Physiol Biophys 39:135–144. https://doi.org/10.4149/gpb_2019054
Nelson A (2006) Co-refinement of multiple-contrast neutron/X-ray reflectivity data using MOTOFIT. J Appl Crystallogr 39:273–276. https://doi.org/10.1107/S0021889806005073
NIH National Institute on Aging (2020) Alzheimers. In: What Is Alzheimer’s Dis. https://www.nia.nih.gov/health/what-alzheimers-disease
NIST Center for Neutron Research (2020) Scattering length density calculator. https://www.ncnr.nist.gov/resources/sldcalc.html
Parratt LG (1954) Surface studies of solids by total reflection of X-rays. Phys Rev 95:359–369. https://doi.org/10.1103/PhysRev.95.359
Pettersen EF, Goddard TD, Huang CC et al (2004) UCSF Chimera—a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612. https://doi.org/10.1002/jcc.20084
Poger D, Mark AE (2012) Lipid bilayers: the effect of force field on ordering and dynamics. J Chem Theory Comput 8:4807–4817. https://doi.org/10.1021/ct300675z
Robinson M, Turnbull S, Lee B, Leonenko Z (2020) The effects of melatonin, serotonin, tryptophan and NAS on the biophysical properties of DPPC monolayers. Biochim Biophys Acta Biomembr 1862:183363. https://doi.org/10.1016/j.bbamem.2020.183363
Róg T, Pasenkiewicz-Gierula M, Vattulainen I, Karttunen M (2009) Ordering effects of cholesterol and its analogues. Biochim Biophys Acta Biomembr 1788:97–121. https://doi.org/10.1016/j.bbamem.2008.08.022
Rondelli V, Brocca P, Motta S et al (2016) Amyloidβ Peptides in interaction with raft-mime model membranes: a neutron reflectivity insight. Sci Rep 6:1–11. https://doi.org/10.1038/srep20997
Rubin AB (2004) Biophysics, vol 1. Nauka, Moscow
Sachs JN, Petrache HI, Woolf TB (2003) Interpretation of small angle X-ray measurements guided by molecular dynamics simulations of lipid bilayers. Chem Phys Lipids 126:211–223. https://doi.org/10.1016/j.chemphyslip.2003.08.001
Schrödinger, LLC (2015) The {PyMOL} molecular graphics system, version ~ 1.8. LLC
Stillwell W (2016a) Chapter 18—membrane-associated processes. In: Stillwell W (ed) An introduction to biological membranes, 2nd edn. Elsevier, pp 381–421
Stillwell W (2016b) Chapter 22—membrane-associated diseases. In: Stillwell W (ed) An introduction to biological membranes, 2nd edn. Elsevier, pp 499–519
Touitou Y (2001) Human aging and melatonin. Clinical relevance. Exp Gerontol 36:1083–1100. https://doi.org/10.1016/S0531-5565(01)00120-6
Verdier Y, Zarándi M, Penke B (2004) Amyloid β-peptide interactions with neuronal and glial cell plasma membrane: binding sites and implications for Alzheimer’s disease. J Pept Sci 10:229–248. https://doi.org/10.1002/psc.573
Xia X, Jiang Q, McDermott J, Han J-DJ (2018) Aging and Alzheimer’s disease: comparison and associations from molecular to system level. Aging Cell 17:e12802–e12802. https://doi.org/10.1111/acel.12802
Yeagle PL (1985) Cholesterol and the cell membrane. Biochim Biophys Acta 822:267–287. https://doi.org/10.1016/0304-4157(85)90011-5
Acknowledgements
The authors acknowledge the access to the experimental and computational facilities of Joint Institute for Nuclear Research.
Funding
This work has been supported by the Russian Science Foundation under Grant 19-72-20186.
Author information
Authors and Affiliations
Contributions
PH, TK and NK designed the experiments that were executed by PH and OT, and matched by MD simulations performed by ED and KK. All the authors approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Availability of data and material
The data are available upon request.
Code availability
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hrubovčák, P., Dushanov, E., Kondela, T. et al. Reflectometry and molecular dynamics study of the impact of cholesterol and melatonin on model lipid membranes. Eur Biophys J 50, 1025–1035 (2021). https://doi.org/10.1007/s00249-021-01564-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-021-01564-y