Summary
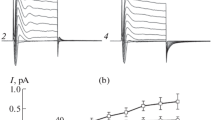
Outwardly rectifying Cl− channels in cultured human Jurkat T-lymphocytes were activated by excising a patch of membrane using the inside-out (i/o) patch-clamp configuration and holding at depolarized voltages for prolonged periods of time (1–6 min at +80 mV, 20°C). The single-channel current at +80 mV was 4.5 ± 0.3 pA and at −80 mV, it was 1.0 ± 0.4 pA. After activation, the probability of being open (P 0)for the lymphocyte channel was voltage independent. Activation of the Cl− channel in lymphocytes was temperature dependent. Nineteen percent of i/o recordings from lymphocytes made at 20°C exhibited Cl− channel activity. In contrast, 49% of recordings made at 30°C showed channel activity. The number of channels in an active patch was not significantly different at the two temperatures. Channel activation in excised, depolarized patches also occurred 20-fold faster at 30°C than at 20°C. There was no marked change in the single-channel conductance at 30°C. Open-channel conductance was blocked by 200 μm indanyloxyacetic acid (IAA) or 1 mm SITS when applied to the intracellular side of the patch. The characteristics of this channel are similar to epithelial outwardly rectifying Cl− channels thought to be involved in fluid secretion
Similar content being viewed by others
References
Artalejo, C.R., Dahmer, M.K., Perlman, R.L., Fox, A.P. 1990. Activation of facilitation calcium channels in chromaffin cells by D1, dopamine receptors through a cAMP/protein kinase A- dependent mechanism. Nature 348:239–242
Artalejo, C.R., Dahmer, M.K., Perlman, R.L., Fox, A.P. 1991. Two types of Ca2+ currents are found in bovine chromaffin cells: Facilitation is due to the recruitment of one type. J. Physiol 432:681–707
Bear, C.E. 1988. Phosphorylation-activated chloride channels in human skin fibroblasts. FEBS Lett 237:145–149
Bridges, R.J., Worrell, R.T., Frizzell, R.A., Benos, D.J. 1989. Stilbene disulfonate blockade of colonic secretory Cl− channels in planar lipid bilayers. Am. J. Physiol 256:C902-C912
Cahalan, M.D., Lewis, R.S. 1988. Role of potassium and chloride channels in volume regulation by T lymphocytes. In: Cell Physiology of Blood. R.B. Gunn and J.C. Parker, editors. pp. 282–301. Rockefeller University Press, New York
Chen, J.H., Schulman, H., Gardner, P. 1989. A cAMP-regulated chloride channel in lymphocytes that is affected in cystic fibrosis. Science 243:657–660
Cliff, W.H., Frizzell, R.A. 1990. Separate Cl− conductances activated by cAMP and Ca− in Cl−-secreting epithelial cells. Prac. Natl. Acad. Sci. USA 87:4956–4960
Cozens, A.L., Yezzi, M.J., Chin, L., Simon, E.M., Friend. D.S., Garber, S.S., Gruenert, D.C. 1992. Chloride ion transport in transformed human airway epithelial cells. In Vitro (in press)
Frizzell, R.A., Halm, D.R. 1990. Chloride channels in epithelial cells. In: Current Topics in Membranes and Transport: Channels and Noise in Epithelial Tissues. S.I. Helman and W. Van Driessche, editors. Vol. 37, pp. 247–282. Academic, New York
Garber, S.S. 1990. Regulation of outwardly rectifying Cl− channels from Jurkat lymphocytes. Proceedings of the Italian Cystic Fibrosis Conference. Sestri Levante, Italy
Gögelein, H. 1988. Chloride channels in epithelia. Biochim. Biophys. Acta 947:521–547
Greger, R., Kunzelmann, K., Gerlach, L. 1989. Mechanisms of chloride transport in secretory epithelia. Ann. NY Acad. Sci 574:403–415
Halm, D.R., Rechkemmer, G., Schoumacher, R.A., Frizzell, R.A. 1988a. Biophysical properties of a chloride channel in the apical membrane of a secretory epithelial cell. Comp. Biochem. Physiol [a] 90:597–601
Halm, D.R., Rechkemmer, G.R., Schoumacher, R.A., Frizzell, R.A. 1988b. Apical membrane chloride channels in a colonic cell line activated by secretory agonists. Am. J. Physiol 254:C505-C511
Hamill, O.P., Marly, A., Neher, E., Sakmann, B., Sigworth, F.J. 1981. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pfluegers Arch 391:85–100
Hanrahan, J.W., Tabcharani, J.A. 1989. Possible role of out-wardly rectifying anion channels in epithelial transport. Ann. NY Acad. Sci 574:30–43
Hayslett, J.P., Gogelein, H., Kunzelmann, K., Greger, R. 1987. Characteristics of apical chloride channels in human colon cells (HT29). Pfluegers Arch 410:487–494
Hoshi, T., Aldrich, R.W. 1988a. Gating kinetics of four classes of voltage-dependent K− channels in pheochromocytoma cells. J. Gen. Physiol 91:107–131
Hoshi, T., Aldrich, R.W. 1988b. Voltage-dependent K+ currents and underlying single K− channels in pheochromocytoma cells. J. Gen. Physiol 91:73–106
Hoshi, T., Smith, S.J. 1987. Large depolarization induces long opening of voltage-dependent calcium channels in adrenal chromaffin cells. J. Neurosci 7:571–580
Hwang, T.-C., Lu, L., Zeitlin, P.L., Gruenert, D.C., Huganir, R., Guggino, W.B. 1989. Cl− channels in CF: Lack of activation by protein kinase C and cAMP-dependent protein kinase. Science 244:1351–1353
Jan, L.Y., Jan, N.J. 1990. How might the diversity of potassium channels be generated? Trends Neurosci 13:415–419
Kunzelmann, K., Tilmann, M., Greger, R. 1990. Cylosolic inhibition of chloride channels. Proceedings of the Italian Cystic Fibrosis Conference. Sestri Levante, Italy
Landry, D.W., Akabas, M.H., Redhead, C., Edelman, A., Cragoe, E.J., Jr., Al-Awqati, Q. 1989. Purification and reconstitution of chloride channels from kidney and trachea. Science 244:1469–1472
Landry, D.W., Reitman, M., Cragoe, E.J., Al-Awqati, Q. 1987. Epithelial chloride channel: Development of inhibitory ligands. J. Gen. Physiol 90:779–798
Li, M., McCann, J.D., Anderson, M.P., Clancy, J.P., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh, M.J. 1989. Regulation of chloride channels by protein kinase C in normal and cystic fibrosis airway epithelia. Science 244:1353–1356
Li, M., McCann, J.D., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh, M.J. 1988. Cyclic AMP-dependent protein kinase opens chloride channels in normal but not cystic fibrosis air-way epithelium. Nature 331:358–360
Lin, P., Gruenstein, E. 1987. Identification of a defective cAMP-stimulated Cl− channel in cystic fibrosis fibroblasts. J. Biol. Chem 262:15345–15347
Pahapill, P.A., Schlichter, L.C. 1990. Kinase-and temperature-regulated chloride channels in normal human T-lymphocytes. Biophys. J 57:394a
Schoumacher, R.A., Shoemaker, R.L., Halm, D.R., Tallant, E.A., Wallace, R.W., Frizzell, R.A. 1987. Phosphorylation fails to activate chloride channels from cystic fibrosis airway cells. Nature 330:752–754
Sole, C.K., Wine, J.J. 1991. Swelling-induced and depolarization-induced Cl− channels in normal and cystic fibrosis epithelial cells. Am. J. Physiol 261:C658-C674
Walsh, K.B., Begenisich, T.B., Kass, R.S. 1989. Beta-adrenergic modulation of cardiac ion channels. Differential temperature sensitivity of potassium and calcium currents. J. Gen. Physiol 93:841–854
Welsh, M.J., Li, M., McCann, J.D. 1989. Activation of normal and cystic fibrosis Cl− channels by voltage, temperature, and trypsin. J. Clin. Invest 84:2002–2007
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garber, S.S. Outwardly rectifying chloride channels in lymphocytes. J. Membarin Biol 127, 49–56 (1992). https://doi.org/10.1007/BF00232757
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232757