Abstract
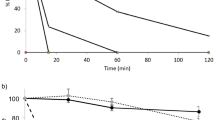
The relative effectiveness of two β-cyclodextrin derivatives, i.e., dimethyl-β-cyclodextrin (DMβCD) and hydroxypropyl-β-cyclodextrin (HPβCD), in enhancing enteral absorption of insulin was evaluated in the lower jejunal/upper ileal segments of the rat by means of an in situ closed loop method. The incorporation of 10% (w/v) DMβCD to a 0.5 mg/ml porcine-zinc insulin solution dramatically increased insulin bioavailability from a negligible value (~0.06%) to 5.63%, when administered enterally at a dose of 20 U/kg. However, addition of 10% (w/v) HPβCD did not improve enteral insulin uptake significantly with a bioavailability of only 0.07%. Similarly, the pharmacodynamic relative efficacy values obtained after the enteral administration of 20 U/kg insulin, 20 U/kg insulin with 10% HPβCD, and 20 U/kg insulin with 10% DMβCD were 0.24%, 0.26%, and 1.75%, respectively. Biodegradation studies of 0.5 mg/ml insulin hexamers by 0.5 µM α-chymotrypsin revealed no inhibitory effect on the enzymatic activity by the two cyclodextrins. On the contrary, the apparent first-order rate constant increased significantly in the presence of 10% DMβCD, suggesting insulin oligomer dissociation by DMβCD. Histopathological examination of the rat intestine was performed to detect tissue damage following enteral administration of the β-cyclodextrin derivatives. Light microscopic inspection indicated no observable tissue damage, thereby arguing direct membrane fluidization as the primary mechanism for enhanced insulin uptake. This study indicates the feasibility of using cyclodextrins as mucosal absorption promoters of proteins and peptide drugs.
Similar content being viewed by others
REFERENCES
U. Derewenda, Z. S. Derewenda, G. G. Dodson, and R. E. Hubbard. Insulin structure. In P. Cuatrecasas and S. Jacobs (eds.), Insulin, Springer-Verlag, Berlin, Heidelberg, New York, London, Paris, Tokyo, Hongkong, 1990, pp. 23–39.
R. J. Schilling and A. K. Mitra. Degradation of insulin by trypsin and alpha-chymotrypsin. Pharm. Res., 8:721–727 (1991).
E. L. Smith, R. L. Hill, and A. Borman. Activity of insulin degraded by leucine aminopeptidase. Biochem. Biophys. Acta., 29:207–208 (1958).
W. A. Banks and A. J. Kastin. Peptides and the blood-brain barrier: Lipophilicity as a predictor of permeability. Brain. Res. Bull., 15:287–292 (1985).
R. J. Schilling and A. K. Mitra. Intestinal mucosal transport of insulin. Int. J. Pharm. 62:53–64 (1990).
M. Morishita, I. Morishita, K. Takayama, Y. Machida, and T. Nagai. Novel oral microspheres of insulin with protease inhibitor protecting from enzymatic degradation. Int. J. Pharm. 78:1–7 (1992).
E. Touitou and A. Rubinstein. Targeted enteral delivery of insulin to rats. Int. J. Pharm. 30:95–99 (1986).
Z. Shao, R. Krishnamoorthy, and A. K. Mitra. Cyclodextrins as nasal absorption promoters of insulin: Mechanistic evaluations. Pharm. Res. 9:1157–1163 (1992).
F. W. H. M. Merkus, J. C. Verhoef, S. G. Romeijn, and N. G. M. Schipper. Absorption enhancing effects of cyclodextrins on intranasally administered insulin in rats. Pharm. Res. 8:588–592 (1991).
N. G. M. Schipper, S. G. Romeijn, J. C. Verhoef, and F. W. H. M. Merkus. Nasal insulin delivery with dimethyl-β-cyclodextrin as an absorption enhancer in rabbits: Powder more effective than liquid formulations. Pharm. Res. 10:682–686 (1993).
T. Irie, K. Wakamatsu, H. Arima, H. Aritomi, and K. Uekama. Enhancing effects of cyclodextrins on nasal absorption of insulin in rats. Int. J. Pharm. 84:129–139 (1992).
Y. Watanabe, Y. Matsumoto, M. Seki, M. Takase, and M. Matsumoto. Absorption enhancement of polypeptide drugs by cyclodextrins. I. Enhanced rectal absorption of insulin from hollow-type suppositories containing insulin and cyclodextrins in rabbits. Chem. Pharm. Bull. 40:3042–3047 (1992).
Y. Watanabe, Y. Matsumoto, K. Kawamoto, S. Yazawa, and M. Matsumoto. Enhancing effect of cyclodextrins on nasal absorption of insulin and its duration in rabbits. Chem. Pharm. Bull. 40:3100–3104 (1992).
Y. Li, Z. Shao, and A. K. Mitra. Dissociation of insulin oligomers by bile salt micelles and its effect on alpha-chymotrypsin-mediated proteolytic degradation. Pharm. Res. 9:864–869 (1992).
B. J. Aungst, N. J. Rogers, and E. Shefter. Comparison of nasal, rectal, buccal, sublingual and intramuscular insulin efficacy and the effects of a bile salt absorption promoter. J. Pharmacol. Exp. Ther., 244:23–27 (1988).
Y. Li and A. K. Mitra. A simple method of correlating pharmacodynamic equivalence with absolute bioavailability following nonparenteral delivery of insulin. Pharm. Res., In press (1994).
R. J. Schilling. Intestinal Mucosal Transport and Metabolism of Insulin, Ph.D. Thesis, Purdue University, West Lafayette, 1991, pp. 1–265.
R. J. Schilling and A. K. Mitra. Pharmacodynamics of insulin following intravenous and enteral administrations of porcinezinc insulin to rats. Pharm. Res. 9:1003–1009 (1992).
K. Matsuyama, S. El-Gizway, and J. H. Perrin. Thermodynamics of binding of aromatic amino acids to α-, β-, and γ-cyclo-dextrins. Drug. Dev. Ind. Pharm. 13:2687–2691 (1987).
J. Szejtli. Cyclodextrins in drug formulations: Part II. Pharm. Tech. 15(8):24–38 (1991).
J. Szejtli. Cyclodextrin Technology, Kluwer Academic Publishers, Dordrecht, Boston, London, 1988, pp. 1–450.
Y. Ohtani, T. Irie, K. Uekama, K. Fukunaga, and J. Pitha. Differential effects of α-, β-, and γ-cyclodextrins on human erythrocytes. Eur. J. Biochem. 186:17–22 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shao, Z., Li, Y., Chermak, T. et al. Cyclodextrins as Mucosal Absorption Promoters of Insulin. II. Effects of β-Cyclodextrin Derivatives on α-Chymotryptic Degradation and Enteral Absorption of Insulin in Rats. Pharm Res 11, 1174–1179 (1994). https://doi.org/10.1023/A:1018997101542
Issue Date:
DOI: https://doi.org/10.1023/A:1018997101542