Abstract
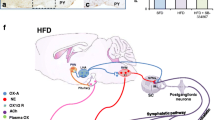
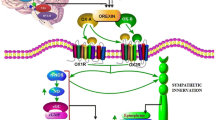
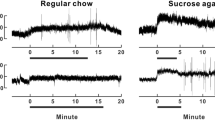
In this review, we focus on the role of orexin signaling in blood pressure control and its potential link to hypertension by summarizing evidence from several experimental animal models of hypertension. Studies using the spontaneously hypertensive rat (SHR) animal model of human essential hypertension show that pharmacological blockade of orexin receptors reduces blood pressure in SHRs but not in Wistar–Kyoto rats. In addition, increased activity of the orexin system contributes to elevated blood pressure and sympathetic nerve activity (SNA) in dark-active period Schlager hypertensive (BPH/2J) mice, another genetic model of neurogenic hypertension. Similar to these two models, Sprague-Dawley rats with stress-induced hypertension display an overactive central orexin system. Furthermore, upregulation of the orexin receptor 1 increases firing of hypothalamic paraventricular nucleus neurons, augments SNA, and contributes to hypertension in the obese Zucker rat, an animal model of obesity-related hypertension. Finally, we propose a hypothesis for the implication of the orexin system in salt-sensitive hypertension. All of this evidence, coupled with the important role of elevated SNA in increasing blood pressure, strongly suggests that hyperactivity of the orexin system contributes to hypertension.

Similar content being viewed by others
References
Amin MS, Reza E, Wang H, Leenen FH (2009) Sodium transport in the choroid plexus and salt-sensitive hypertension. Hypertension 54:860–867. doi:10.1161/hypertensionaha.108.125807
Andresen MC, Kuraoka S, Brown AM (1980) Baroreceptor function and changes in strain sensitivity in normotensive and spontaneously hypertensive rats. Circ Res 47:821–828
Antunes VR, Brailoiu GC, Kwok EH, Scruggs P, Dun NJ (2001) Orexins/hypocretins excite rat sympathetic preganglionic neurons in vivo and in vitro. Am J Physiol Regul Integr Comp Physiol 281:R1801–R1807
Backberg M, Hervieu G, Wilson S, Meister B (2002) Orexin receptor-1 (OX-R1) immunoreactivity in chemically identified neurons of the hypothalamus: focus on orexin targets involved in control of food and water intake. Eur J Neurosci 15:315–328
Baldo BA, Daniel RA, Berridge CW, Kelley AE (2003) Overlapping distributions of orexin/hypocretin- and dopamine-beta-hydroxylase immunoreactive fibers in rat brain regions mediating arousal, motivation, and stress. J Comp Neurol 464:220–237. doi:10.1002/cne.10783
Carlson SH, Shelton J, White CR, Wyss JM (2000) Elevated sympathetic activity contributes to hypertension and salt sensitivity in diabetic obese Zucker rats. Hypertension 35:403–408
Carrive P (2013) Orexin, orexin receptor antagonists and central cardiovascular control. Front Neurosci 7:257. doi:10.3389/fnins.2013.00257
Chen CT, Hwang LL, Chang JK, Dun NJ (2000) Pressor effects of orexins injected intracisternally and to rostral ventrolateral medulla of anesthetized rats. Am J Physiol Regul Integr Comp Physiol 278:R692–R697
Ciriello J, de Oliveira CV (2003) Cardiac effects of hypocretin-1 in nucleus ambiguus. Am J Physiol Regul Integr Comp Physiol 284:R1611–R1620. doi:10.1152/ajpregu.00719.2002
Ciriello J, Li Z, de Oliveira CV (2003) Cardioacceleratory responses to hypocretin-1 injections into rostral ventromedial medulla. Brain Res 991:84–95
Clifford L, Dampney BW, Carrive P (2015) Spontaneously hypertensive rats have more orexin neurons in their medial hypothalamus than normotensive rats. Exp Physiol 100:388–398. doi:10.1113/expphysiol.2014.084137
Cluderay JE, Harrison DC, Hervieu GJ (2002) Protein distribution of the orexin-2 receptor in the rat central nervous system. Regul Pept 104:131–144
Date Y et al (1999) Orexins, orexigenic hypothalamic peptides, interact with autonomic, neuroendocrine and neuroregulatory systems. Proc Natl Acad Sci USA 96:748–753
Date Y, Mondal MS, Matsukura S, Nakazato M (2000) Distribution of orexin-A and orexin-B (hypocretins) in the rat spinal cord. Neurosci Lett 288:87–90
Davern PJ, Nguyen-Huu TP, La Greca L, Abdelkader A, Head GA (2009) Role of the sympathetic nervous system in Schlager genetically hypertensive mice. Hypertension 54:852–859. doi:10.1161/hypertensionaha.109.136069
de Lecea L (2012) Hypocretins and the neurobiology of sleep-wake mechanisms. Prog Brain Res 198:15–24. doi:10.1016/b978-0-444-59489-1.00003-3
de Lecea L et al (1998) The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci USA 95:322–327
Dias MB, Li A, Nattie EE (2009) Antagonism of orexin receptor-1 in the retrotrapezoid nucleus inhibits the ventilatory response to hypercapnia predominantly in wakefulness. J Physiol 587:2059–2067. doi:10.1113/jphysiol.2008.168260
Dias MB, Li A, Nattie E (2010) The orexin receptor 1 (OX1R) in the rostral medullary raphe contributes to the hypercapnic chemoreflex in wakefulness, during the active period of the diurnal cycle. Respir Physiol Neurobiol 170:96–102. doi:10.1016/j.resp.2009.12.002
Follwell MJ, Ferguson AV (2002) Cellular mechanisms of orexin actions on paraventricular nucleus neurones in rat hypothalamus. J Physiol 545:855–867
Furlong TM, Vianna DM, Liu L, Carrive P (2009) Hypocretin/orexin contributes to the expression of some but not all forms of stress and arousal. Eur J Neurosci 30:1603–1614. doi:10.1111/j.1460-9568.2009.06952.x
Gordon FJ, Matsuguchi H, Mark AL (1981) Abnormal baroreflex control of heart rate in prehypertensive and hypertensive Dahl genetically salt-sensitive rats. Hypertension 3:I135–I141
Goto A, Ikeda T, Tobian L, Iwai J, Johnson MA (1981) Brain lesions in the paraventricular nuclei and catecholaminergic neurons minimize salt hypertension in Dahl salt-sensitive rats. Clin Sci (London, England: 1979) 61(Suppl 7):53s–55s
Hahn JD, Swanson LW (2010) Distinct patterns of neuronal inputs and outputs of the juxtaparaventricular and suprafornical regions of the lateral hypothalamic area in the male rat. Brain Res Rev 64:14–103. doi:10.1016/j.brainresrev.2010.02.002
Hervieu GJ, Cluderay JE, Harrison DC, Roberts JC, Leslie RA (2001) Gene expression and protein distribution of the orexin-1 receptor in the rat brain and spinal cord. Neuroscience 103:777–797
Huang BS, Van Vliet BN, Leenen FH (2004) Increases in CSF [Na +] precede the increases in blood pressure in Dahl S rats and SHR on a high-salt diet. Am J Physiol-Heart Circ Physiol 287:H1160–H1166
Hurley SW, Johnson AK (2014) The role of the lateral hypothalamus and orexin in ingestive behavior: a model for the translation of past experience and sensed deficits into motivated behaviors. Front Syst Neurosci 8:216. doi:10.3389/fnsys.2014.00216
Jackson KL et al (2013) A novel interaction between sympathetic overactivity and aberrant regulation of renin by miR-181a in BPH/2J genetically hypertensive mice. Hypertension 62:775–781. doi:10.1161/hypertensionaha.113.01701
Jackson KL, Dampney BW, Moretti JL, Stevenson ER, Davern PJ, Carrive P, Head GA (2016) Contribution of orexin to the neurogenic hypertension in BPH/2J mice. Hypertension 67:959–969. doi:10.1161/hypertensionaha.115.07053
Johnson PL et al (2010) A key role for orexin in panic anxiety. Nat Med 16:111–115. doi:10.1038/nm.2075
Judy WV, Watanabe AM, Henry DP, Besch HR Jr, Murphy WR, Hockel GM (1976) Sympathetic nerve activity: role in regulation of blood pressure in the spontaneously hypertensive rat. Circ Res 38:21–29
Kayaba Y et al (2003) Attenuated defense response and low basal blood pressure in orexin knockout mice. Am J Physiol Regul Integr Comp Physiol 285:R581–R593. doi:10.1152/ajpregu.00671.2002
Kishi T (2013) Regulation of the sympathetic nervous system by nitric oxide and oxidative stress in the rostral ventrolateral medulla: 2012 Academic Conference Award from the Japanese Society of Hypertension. Hypertens Res 36:845–851. doi:10.1038/hr.2013.73
Kunii K, Yamanaka A, Nambu T, Matsuzaki I, Goto K, Sakurai T (1999) Orexins/hypocretins regulate drinking behaviour. Brain Res 842:256–261
Lee YH, Dai YW, Huang SC, Li TL, Hwang LL (2013) Blockade of central orexin 2 receptors reduces arterial pressure in spontaneously hypertensive rats. Exp Physiol 98:1145–1155. doi:10.1113/expphysiol.2013.072298
Lee YH, Tsai MC, Li TL, Dai YW, Huang SC, Hwang LL (2015) Spontaneously hypertensive rats have more orexin neurons in the hypothalamus and enhanced orexinergic input and orexin 2 receptor-associated nitric oxide signalling in the rostral ventrolateral medulla. Exp Physiol 100:993–1007. doi:10.1113/ep085016
Li A, Nattie E (2010) Antagonism of rat orexin receptors by almorexant attenuates central chemoreception in wakefulness in the active period of the diurnal cycle. J Physiol 588:2935–2944. doi:10.1113/jphysiol.2010.191288
Li A, Nattie E (2014) Orexin, cardio-respiratory function, and hypertension. Front Neurosci 8:22. doi:10.3389/fnins.2014.00022
Li A, Hindmarch CC, Nattie EE, Paton JF (2013) Antagonism of orexin receptors significantly lowers blood pressure in spontaneously hypertensive rats. J Physiol 591:4237–4248. doi:10.1113/jphysiol.2013.256271
Li A, Roy SH, Nattie EE (2016) An augmented CO2 chemoreflex and overactive orexin system are linked with hypertension in young and adult spontaneously hypertensive rats. J Physiol 594:4967–4980. doi:10.1113/jp272199
Lu XY, Bagnol D, Burke S, Akil H, Watson SJ (2000) Differential distribution and regulation of OX1 and OX2 orexin/hypocretin receptor messenger RNA in the brain upon fasting. Horm Behav 37:335–344. doi:10.1006/hbeh.2000.1584
Marcus JN, Aschkenasi CJ, Lee CE, Chemelli RM, Saper CB, Yanagisawa M, Elmquist JK (2001) Differential expression of orexin receptors 1 and 2 in the rat brain. J Comp Neurol 435:6–25
Marques FZ, Campain AE, Davern PJ, Yang YH, Head GA, Morris BJ (2011a) Genes influencing circadian differences in blood pressure in hypertensive mice. PLoS ONE 6:e19203. doi:10.1371/journal.pone.0019203
Marques FZ, Campain AE, Davern PJ, Yang YH, Head GA, Morris BJ (2011b) Global identification of the genes and pathways differentially expressed in hypothalamus in early and established neurogenic hypertension. Physiol Genom 43:766–771. doi:10.1152/physiolgenomics.00009.2011
Nakamura K, Cowley AW (1989) Sequential changes of cerebrospinal fluid sodium during the development of hypertension in Dahl rats. Hypertension 13:243–249. doi:10.1161/01.hyp.13.3.243
Nakamura A, Zhang W, Yanagisawa M, Fukuda Y, Kuwaki T (2007) Vigilance state-dependent attenuation of hypercapnic chemoreflex and exaggerated sleep apnea in orexin knockout mice. J Appl Physiol (Bethesda, MD: 1985) 102:241–248. doi:10.1152/japplphysiol.00679.2006
Nambu T, Sakurai T, Mizukami K, Hosoya Y, Yanagisawa M, Goto K (1999) Distribution of orexin neurons in the adult rat brain. Brain Res 827:243–260
Peyron C, Tighe DK, van den Pol AN, de Lecea L, Heller HC, Sutcliffe JG, Kilduff TS (1998) Neurons containing hypocretin (orexin) project to multiple neuronal systems. J Neurosci 18:9996–10015
Sakurai T (2007) The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nat Rev Neurosci 8:171–181. doi:10.1038/nrn2092
Sakurai T (2014) The role of orexin in motivated behaviours. Nat Rev Neurosci 15:719–731. doi:10.1038/nrn3837
Sakurai T et al (1998) Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92:573–585
Samson WK, Taylor MM, Follwell M, Ferguson AV (2002) Orexin actions in hypothalamic paraventricular nucleus: physiological consequences and cellular correlates. Regul Pept 104:97–103
Shahid IZ, Rahman AA, Pilowsky PM (2012) Orexin A in rat rostral ventrolateral medulla is pressor, sympatho-excitatory, increases barosensitivity and attenuates the somato-sympathetic reflex. Br J Pharmacol 165:2292–2303. doi:10.1111/j.1476-5381.2011.01694.x
Shirasaka T, Miyahara S, Kunitake T, Jin QH, Kato K, Takasaki M, Kannan H (2001) Orexin depolarizes rat hypothalamic paraventricular nucleus neurons. Am J Physiol Regul Integr Comp Physiol 281:R1114–R1118
Simchon S, Manger W, Golanov E, Kamen J, Sommer G, Marshall CH (1999) Handling 22NaCl by the blood-brain barrier and kidney: its relevance to salt-induced hypertension in dahl rats. Hypertension 33:517–523
Simms AE, Paton JF, Pickering AE, Allen AM (2009) Amplified respiratory-sympathetic coupling in the spontaneously hypertensive rat: does it contribute to hypertension? J Physiol 587:597–610. doi:10.1113/jphysiol.2008.165902
Takeshita A, Mark AL, Brody MJ (1979) Prevention of salt-induced hypertension in the Dahl strain by 6-hydroxydopamine. Am J Physiol 236:H48–H52
Trivedi P, Yu H, MacNeil DJ, Van der Ploeg LH, Guan XM (1998) Distribution of orexin receptor mRNA in the rat brain. FEBS Lett 438:71–75
Tsujino N, Sakurai T (2009) Orexin/hypocretin: a neuropeptide at the interface of sleep, energy homeostasis, and reward system. Pharmacol Rev 61:162–176. doi:10.1124/pr.109.001321
Williams RH, Jensen LT, Verkhratsky A, Fugger L, Burdakov D (2007) Control of hypothalamic orexin neurons by acid and CO2. Proc Natl Acad Sci USA 104:10685–10690. doi:10.1073/pnas.0702676104
Xiao F et al (2013) Orexin A regulates cardiovascular responses in stress-induced hypertensive rats. Neuropharmacology 67:16–24. doi:10.1016/j.neuropharm.2012.10.021
Yamanaka A et al (2003) Hypothalamic orexin neurons regulate arousal according to energy balance in mice. Neuron 38:701–713
Zheng H, Patterson LM, Berthoud HR (2005) Orexin-A projections to the caudal medulla and orexin-induced c-Fos expression, food intake, and autonomic function. J Comp Neurol 485:127–142. doi:10.1002/cne.20515
Zhou JJ, Yuan F, Zhang Y, Li DP (2015) Upregulation of orexin receptor in paraventricular nucleus promotes sympathetic outflow in obese Zucker rats. Neuropharmacology 99:481–490. doi:10.1016/j.neuropharm.2015.08.012
Acknowledgments
NIHR15HL129213 (Shan); Michigan Technological University Research Excellence Fund (Shan).
Author information
Authors and Affiliations
Contributions
Michael J. Huber wrote the manuscript, with editorial comments from Qing-Hui Chen and Zhiying Shan.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Huber, M.J., Chen, QH. & Shan, Z. The Orexin System and Hypertension. Cell Mol Neurobiol 38, 385–391 (2018). https://doi.org/10.1007/s10571-017-0487-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-017-0487-z