Abstract
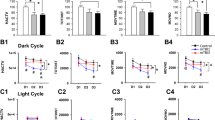
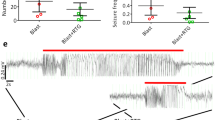
Circadian rhythms modulate many physiologic processes and behaviors. Therefore, their disruption causes a variety of potential adverse effects in humans and animals. Circadian disruption induced by constant light exposure has been discovered to produce pathophysiologic consequences after brain injury. However, the underlying mechanisms that lead to more severe impairment and disruption of neurophysiologic processes are not well understood. Here, we evaluated the effect of constant light exposure on the neurobehavioral impairment and survival of neurons in rats after traumatic brain injury (TBI). Sixty adult male Sprague–Dawley rats were subjected to a weight-drop model of TBI and then exposed to either a standard 12-/12-h light/dark cycle or a constant 24-h light/light cycle for 14 days. Our results showed that 14 days of constant light exposure after TBI significantly worsened the sensorimotor and cognitive deficits, which were associated with decreased body weight, impaired water and food intake, increased cortical lesion volume, and decreased neuronal survival. Furthermore, environmental circadian disruption inhibited cell proliferation and newborn cell survival and decreased immature cell production in rats subjected to the TBI model. We conclude that circadian disruption induced by constant light exposure worsens histologic and neurobehavioral impairment and inhibits neurogenesis in adult TBI rats. Our novel findings suggest that light exposure should be decreased and circadian rhythm reestablished in hospitalized TBI patients and that drugs and strategies that maintain circadian rhythm would offer a novel therapeutic option.






Similar content being viewed by others
Change history
30 June 2020
A Correction to this paper has been published: https://doi.org/10.1007/s10571-020-00913-3
References
Alibhai FJ, Tsimakouridze EV, Reitz CJ, Pyle WG, Martino TA (2015) Consequences of circadian and sleep disturbances for the cardiovascular system. Can J Cardiol 31(7):860–872. doi:10.1016/j.cjca.2015.01.015
Antle MC, Smith VM, Sterniczuk R, Yamakawa GR, Rakai BD (2009) Physiological responses of the circadian clock to acute light exposure at night. Rev Endocr Metab Disord 10(4):279–291. doi:10.1007/s11154-009-9116-6
Aubrecht TG, Jenkins R, Nelson RJ (2014) Dim light at night increases body mass of female mice. Chronobiol Int. doi:10.3109/07420528.2014.986682
Bailey SM, Udoh US, Young ME (2014) Circadian regulation of metabolism. J Endocrinol 222(2):R75–96. doi:10.1530/JOE-14-0200
Bales JW, Macfarlane K, Dixon CE (2012) Vestibular assessments following traumatic brain injury. In: Animal models of acute neurological injuries II. Springer, pp 385–396
Bedrosian TA, Fonken LK, Walton JC, Haim A, Nelson RJ (2011) Dim light at night provokes depression-like behaviors and reduces CA1 dendritic spine density in female hamsters. Psychoneuroendocrinology 36(7):1062–1069. doi:10.1016/j.psyneuen.2011.01.004
Boone DR et al (2012) Traumatic brain injury-induced dysregulation of the circadian clock. PLoS ONE 7(10):e46204. doi:10.1371/journal.pone.0046204
Bouchard-Cannon P, Mendoza-Viveros L, Yuen A, Kaern M, Cheng HY (2013) The circadian molecular clock regulates adult hippocampal neurogenesis by controlling the timing of cell-cycle entry and exit. Cell Rep 5(4):961–973. doi:10.1016/j.celrep.2013.10.037
Castro JP, Frussa-Filho R, Fukushiro DF, Chinen CC, Abilio VC, Silva RH (2005) Effects of long-term continuous exposure to light on memory and anxiety in mice. Physiol Behav 86(1–2):218–223. doi:10.1016/j.physbeh.2005.07.009
Chakir I, Dumont S, Pevet P, Ouarour A, Challet E, Vuillez P (2015) Pineal melatonin is a circadian time-giver for leptin rhythm in Syrian hamsters. Front Neurosci 9:190. doi:10.3389/fnins.2015.00190
Chang CF, Cho S, Wang J (2014) (-)-Epicatechin protects hemorrhagic brain via synergistic Nrf2 pathways. Ann Clin Transl Neurol 1(4):258–271. doi:10.1002/acn3.54
Cheng T et al (2015) Wharton’s Jelly transplantation improves neurologic function in a rat model of traumatic brain injury. Cell Mol Neurobiol. doi:10.1007/s10571-015-0159-9
Colwell CS (2011) Linking neural activity and molecular oscillations in the SCN. Nat Rev Neurosci 12(10):553–569. doi:10.1038/nrn3086
Coomans CP et al (2013) Detrimental effects of constant light exposure and high-fat diet on circadian energy metabolism and insulin sensitivity. FASEB J 27(4):1721–1732. doi:10.1096/fj.12-210898
Duclos C et al (2014) Sleep and wake disturbances following traumatic brain injury. Pathol Biol (Paris) 62(5):252–261
Fonken LK, Aubrecht TG, Melendez-Fernandez OH, Weil ZM, Nelson RJ (2013a) Dim light at night disrupts molecular circadian rhythms and increases body weight. J Biol Rhythms 28(4):262–271. doi:10.1177/0748730413493862
Fonken LK, Weil ZM, Nelson RJ (2013b) Mice exposed to dim light at night exaggerate inflammatory responses to lipopolysaccharide. Brain Behav Immun 34:159–163. doi:10.1016/j.bbi.2013.08.011
Fonseca Costa SS, Ripperger JA (2015) Impact of the circadian clock on the aging process. Front Neurol. doi:10.3389/fneur.2015.00043
Giebultowicz JM, Long DM (2015) Ageing and circadian rhythms. Curr Opin Insect Sci 7:82–86. doi:10.1016/j.cois.2015.03.001
Hardin KA (2009) Sleep in the ICU: potential mechanisms and clinical implications. Chest J 136(1):284–294
Hunter A, Hatcher J, Virley D, Nelson P, Irving E, Hadingham S, Parsons A (2000) Functional assessments in mice and rats after focal stroke. Neuropharmacology 39(5):806–816
Kott J, Leach G, Yan L (2012) Direction-dependent effects of chronic “jet-lag” on hippocampal neurogenesis. Neurosci Lett 515(2):177–180. doi:10.1016/j.neulet.2012.03.048
Ma WP, Cao J, Tian M, Cui MH, Han HL, Yang YX, Xu L (2007) Exposure to chronic constant light impairs spatial memory and influences long-term depression in rats. Neurosci Res 59(2):224–230. doi:10.1016/j.neures.2007.06.1474
Martinez-Vargas M et al (2012) Sleep deprivation has a neuroprotective role in a traumatic brain injury of the rat. Neurosci Lett 529(2):118–122. doi:10.1016/j.neulet.2012.09.037
Matulka K, Lin HH, Hribkova H, Uwanogho D, Dvorak P, Sun YM (2013) PTP1B is an effector of activin signaling and regulates neural specification of embryonic stem cells. Cell Stem Cell 13(6):706–719. doi:10.1016/j.stem.2013.09.016
Miyamoto H, Nakamaru-Ogiso E, Hamada K, Hensch TK (2012) Serotonergic integration of circadian clock and ultradian sleep-wake cycles. J Neurosci 32(42):14794–14803. doi:10.1523/jneurosci.0793-12.2012
Mohawk JA, Green CB, Takahashi JS (2012) Central and peripheral circadian clocks in mammals. Annu Rev Neurosci 35:445–462. doi:10.1146/annurev-neuro-060909-153128
Mueller AD, Mear RJ, Mistlberger RE (2011) Inhibition of hippocampal neurogenesis by sleep deprivation is independent of circadian disruption and melatonin suppression. Neuroscience 193:170–181. doi:10.1016/j.neuroscience.2011.07.019
Panda S et al (2002) Melanopsin (Opn4) requirement for normal light-induced circadian phase shifting. Science (New York, NY) 298(5601):2213–2216. doi:10.1126/science.1076848
Park SY, Walker JJ, Johnson NW, Zhao Z, Lightman SL, Spiga F (2013) Constant light disrupts the circadian rhythm of steroidogenic proteins in the rat adrenal gland. Mol Cell Endocrinol 371(1–2):114–123. doi:10.1016/j.mce.2012.11.010
Roenneberg T, Kantermann T, Juda M, Vetter C, Allebrandt KV (2013) Light and the human circadian clock. Handb Exp Pharmacol 217:311–331. doi:10.1007/978-3-642-25950-0_13
Stevens RG et al (2007) Meeting report: the role of environmental lighting and circadian disruption in cancer and other diseases. Environ Health Perspect 115(9):1357–1362. doi:10.1289/ehp.10200
Stevens RG, Brainard GC, Blask DE, Lockley SW, Motta ME (2013) Adverse health effects of nighttime lighting: comments on American Medical Association policy statement. Am J Prev Med 45(3):343–346. doi:10.1016/j.amepre.2013.04.011
Stoica BA, Faden AI (2010) Cell death mechanisms and modulation in traumatic brain injury. Neurotherapeutics 7(1):3–12
Sun D, Daniels TE, Rolfe A, Waters M, Hamm R (2015) Inhibition of injury-induced cell proliferation in the dentate gyrus of the hippocampus impairs spontaneous cognitive recovery after traumatic brain injury. J Neurotrauma 32(7):495–505. doi:10.1089/neu.2014.3545
Tataroglu O, Aksoy A, Yilmaz A, Canbeyli R (2004) Effect of lesioning the suprachiasmatic nuclei on behavioral despair in rats. Brain Res 1001(1–2):118–124. doi:10.1016/j.brainres.2003.11.063
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1(2):848–858
Wang F et al (2002) Inhibition of cyclin-dependent kinases improves CA1 neuronal survival and behavioral performance after global ischemia in the rat. J Cereb Blood Flow Metab 22(2):171–182
Wang H et al (2014) Intravenous administration of Honokiol provides neuroprotection and improves functional recovery after traumatic brain injury through cell cycle inhibition. Neuropharmacology 86:9–21. doi:10.1016/j.neuropharm.2014.06.018
Wang W et al (2015) Protective effects of chinese herbal medicine rhizoma drynariae in rats after traumatic brain injury and identification of active compound. Mol Neurobiol. doi:10.1007/s12035-015-9385-x
Zelinski EL, Deibel SH, McDonald RJ (2014) The trouble with circadian clock dysfunction: multiple deleterious effects on the brain and body. Neurosci Biobehav Rev 40:80–101. doi:10.1016/j.neubiorev.2014.01.007
Zuurbier LA, Ikram MA, Luik AI, Hofman A, Van Someren EJ, Vernooij MW, Tiemeier H (2015) Cerebral small vessel disease is related to disturbed 24-h activity rhythms: a population-based study. Eur J Neurol. doi:10.1111/ene.12775
Acknowledgments
This study was supported by the National Natural Science Foundation of China (81171177,81471306, U1404313), the Innovative Research Team (in Science and Technology) of the University of Henan Province (15IRTSTHN022), the Plan For Scientific Innovation Talent of Henan Province (154200510008) and NIH (R01NS078026, R01AT007317). T.C. is a recipient of the China Scholarship Council Joint PhD Training award. We thank Claire Levine, MS, ELS, for assistance with this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Dongpeng Li and Shanshan Ma have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, D., Ma, S., Guo, D. et al. Environmental Circadian Disruption Worsens Neurologic Impairment and Inhibits Hippocampal Neurogenesis in Adult Rats After Traumatic Brain Injury. Cell Mol Neurobiol 36, 1045–1055 (2016). https://doi.org/10.1007/s10571-015-0295-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0295-2