Abstract
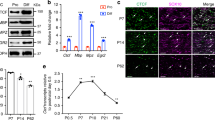
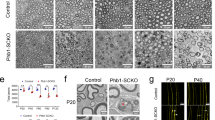
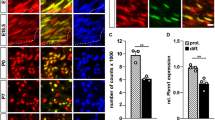
Schwann cell myelination is tightly regulated by timely expression of key transcriptional regulators that respond to specific environmental cues, but the molecular mechanisms underlying such a process are poorly understood. We found that the acetylation state of NF-κB, which is regulated by histone deacetylases (HDACs) 1 and 2, is critical for orchestrating the myelination program. Mice lacking both HDACs 1 and 2 (HDAC1/2) exhibited severe myelin deficiency with Schwann cell development arrested at the immature stage. NF-κB p65 became heavily acetylated in HDAC1/2 mutants, inhibiting the expression of positive regulators of myelination and inducing the expression of differentiation inhibitors. We observed that the NF-κB protein complex switched from associating with p300 to associating with HDAC1/2 as Schwann cells differentiated. NF-κB and HDAC1/2 acted in a coordinated fashion to regulate the transcriptionally linked chromatin state for Schwann cell myelination. Thus, our results reveal an HDAC-mediated developmental switch for controlling myelination in the peripheral nervous system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Suter, U. & Scherer, S.S. Disease mechanisms in inherited neuropathies. Nat. Rev. Neurosci. 4, 714–726 (2003).
Jessen, K.R. & Mirsky, R. The origin and development of glial cells in peripheral nerves. Nat. Rev. Neurosci. 6, 671–682 (2005).
Svaren, J. & Meijer, D. The molecular machinery of myelin gene transcription in Schwann cells. Glia 56, 1541–1551 (2008).
Shahbazian, M.D. & Grunstein, M. Functions of site-specific histone acetylation and deacetylation. Annu. Rev. Biochem. 76, 75–100 (2007).
Ye, F. et al. HDAC1 and HDAC2 regulate oligodendrocyte differentiation by disrupting the beta-catenin–TCF interaction. Nat. Neurosci. 12, 829–838 (2009).
Shen, S. et al. Age-dependent epigenetic control of differentiation inhibitors is critical for remyelination efficiency. Nat. Neurosci. 11, 1024–1034 (2008).
Jaegle, M. et al. The POU proteins Brn-2 and Oct-6 share important functions in Schwann cell development. Genes Dev. 17, 1380–1391 (2003).
Nickols, J.C., Valentine, W., Kanwal, S. & Carter, B.D. Activation of the transcription factor NF-κB in Schwann cells is required for peripheral myelin formation. Nat. Neurosci. 6, 161–167 (2003).
Calao, M., Burny, A., Quivy, V., Dekoninck, A. & Van Lint, C. A pervasive role of histone acetyltransferases and deacetylases in an NF-κB signaling code. Trends Biochem. Sci. 33, 339–349 (2008).
Chen, L.F., Mu, Y. & Greene, W.C. Acetylation of RelA at discrete sites regulates distinct nuclear functions of NF-κB. EMBO J. 21, 6539–6548 (2002).
Rothgiesser, K.M., Fey, M. & Hottiger, M.O. Acetylation of p65 at lysine 314 is important for late NF-κB–dependent gene expression. BMC Genomics 11, 22 (2010).
Martin, C. & Zhang, Y. The diverse functions of histone lysine methylation. Nat. Rev. Mol. Cell Biol. 6, 838–849 (2005).
Strahl, B.D. & Allis, C.D. The language of covalent histone modifications. Nature 403, 41–45 (2000).
Antonellis, A. et al. Identification of neural crest and glial enhancers at the mouse Sox10 locus through transgenesis in zebrafish. PLoS Genet. 4, e1000174 (2008).
He, Y. et al. Yy1 as a molecular link between neuregulin and transcriptional modulation of peripheral myelination. Nat. Neurosci. 13, 1472–1480 (2010).
Fancy, S.P. et al. Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Genes Dev. 23, 1571–1585 (2009).
Jacob, C. et al. HDAC1 and HDAC2 control the transcriptional program of myelination and the survival of Schwann cells. Nat. Neurosci. published online, doi:10.1038/nn.2762 (20 March 2011).
Harada, N. et al. Intestinal polyposis in mice with a dominant stable mutation of the beta-catenin gene. EMBO J. 18, 5931–5942 (1999).
Chen, Y. et al. The oligodendrocyte-specific G protein–coupled receptor GPR17 is a cell-intrinsic timer of myelination. Nat. Neurosci. 12, 1398–1406 (2009).
Brockes, J.P., Fields, K.L. & Raff, M.C. Studies on cultured rat Schwann cells. I. Establishment of purified populations from cultures of peripheral nerve. Brain Res. 165, 105–118 (1979).
Yeiser, E.C., Rutkoski, N.J., Naito, A., Inoue, J. & Carter, B.D. Neurotrophin signaling through the p75 receptor is deficient in traf6−/− mice. J. Neurosci. 24, 10521–10529 (2004).
Jang, S.W., LeBlanc, S.E., Roopra, A., Wrabetz, L. & Svaren, J. In vivo detection of Egr2 binding to target genes during peripheral nerve myelination. J. Neurochem. 98, 1678–1687 (2006).
Bernstein, B.E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Acknowledgements
We would like to thank D. Meijer for Dhh-cre mice, B. Carter and J. Chan for critical comments, and P. Casaccia for initial discussions. We thank Q. Weng and Z. Ma for technical support, O. Nakagawa and J. Chen for the p300/CBP and p65/RelA expression vectors, and R. Kageyama for the Hes5 luciferase reporter. This study was funded in part by grants from the US National Institutes of Health (NS072427) and the National Multiple Sclerosis Society (RG3978) to Q.R.L.
Author information
Authors and Affiliations
Contributions
Y.C. conducted the majority of the experiments and analyzed the data. H.W. and X.X. contributed to HDAC mutant generation, phenotype analysis and biochemical assays. S.O.K., J.S. and H.A.S. provided reagents and input. M.H. provided the p65 mutant–expression vectors. K.A.N. provided CNP-Cre mice for initial phenotype observation. E.N.O. provided loxP-flanked HDAC1 and HDAC2 mice and inputs. Q.R.L. supervised the project, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1400 kb)
Rights and permissions
About this article
Cite this article
Chen, Y., Wang, H., Yoon, S. et al. HDAC-mediated deacetylation of NF-κB is critical for Schwann cell myelination. Nat Neurosci 14, 437–441 (2011). https://doi.org/10.1038/nn.2780
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2780
This article is cited by
-
LPA1 signaling drives Schwann cell dedifferentiation in experimental autoimmune neuritis
Journal of Neuroinflammation (2021)
-
Myelin Biology
Neurotherapeutics (2021)
-
Aberrant DNA methylation profile exacerbates inflammation and neurodegeneration in multiple sclerosis patients
Journal of Neuroinflammation (2020)
-
CTCF-mediated chromatin looping in EGR2 regulation and SUZ12 recruitment critical for peripheral myelination and repair
Nature Communications (2020)
-
Harnessing the HDAC–histone deacetylase enzymes, inhibitors and how these can be utilised in tissue engineering
International Journal of Oral Science (2019)