Abstract
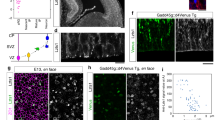
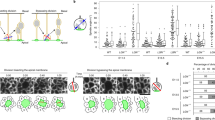
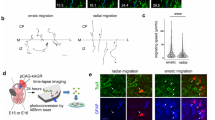
The construction of cerebral cortex begins with the formation of radial glia. Once formed, polarized radial glial cells divide either symmetrically or asymmetrically to balance appropriate production of progenitor cells and neurons. Following birth, neurons use the processes of radial glia as scaffolding for oriented migration. Radial glia therefore provide an instructive structural matrix to coordinate the generation and placement of distinct groups of cortical neurons in the developing cerebral cortex. We found that Arl13b, a cilia-enriched small GTPase that is mutated in Joubert syndrome, was critical for the initial formation of the polarized radial progenitor scaffold. Using developmental stage–specific deletion of Arl13b in mouse cortical progenitors, we found that early neuroepithelial deletion of ciliary Arl13b led to a reversal of the apical–basal polarity of radial progenitors and aberrant neuronal placement. Arl13b modulated ciliary signaling necessary for radial glial polarity. Our findings indicate that Arl13b signaling in primary cilia is crucial for the initial formation of a polarized radial glial scaffold and suggest that disruption of this process may contribute to aberrant neurodevelopment and brain abnormalities in Joubert syndrome–related ciliopathies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Rakic, P. Specification of cerebral cortical areas. Science 241, 170–176 (1988).
Miyata, T., Kawaguchi, A., Okano, H. & Ogawa, M. Asymmetric inheritance of radial glial fibers by cortical neurons. Neuron 31, 727–741 (2001).
Noctor, S.C., Martinez-Cerdeno, V., Ivic, L. & Kriegstein, A.R. Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nat. Neurosci. 7, 136–144 (2004).
Götz, M. & Huttner, W.B. The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 6, 777–788 (2005).
Manzini, M.C. & Walsh, C.A. What disorders of cortical development tell us about the cortex: one plus one does not always make two. Curr. Opin. Genet. Dev. 21, 333–339 (2011).
Tabata, H., Kanatani, S. & Nakajima, K. Differences of migratory behavior between direct progeny of apical progenitors and basal progenitors in the developing cerebral cortex. Cereb. Cortex 19, 2092–2105 (2009).
Valiente, M. & Marin, O. Neuronal migration mechanisms in development and disease. Curr. Opin. Neurobiol. 20, 68–78 (2010).
Yokota, Y. et al. Radial glial dependent and independent dynamics of interneuronal migration in the developing cerebral cortex. PLoS ONE 2, e794 (2007).
Oh, E.C. & Katsanis, N. Cilia in vertebrate development and disease. Development 139, 443–448 (2012).
Cantagrel, V. et al. Mutations in the cilia gene ARL13B lead to the classical form of Joubert syndrome. Am. J. Hum. Genet. 83, 170–179 (2008).
Eggenschwiler, J.T. & Anderson, K.V. Cilia and developmental signaling. Annu. Rev. Cell Dev. Biol. 23, 345–373 (2007).
Hildebrandt, F., Benzing, T. & Katsanis, N. Ciliopathies. N. Engl. J. Med. 364, 1533–1543 (2011).
Lancaster, M.A. & Gleeson, J.G. The primary cilium as a cellular signaling center: lessons from disease. Curr. Opin. Genet. Dev. 19, 220–229 (2009).
Louvi, A. & Grove, E.A. Cilia in the CNS: the quiet organelle claims center stage. Neuron 69, 1046–1060 (2011).
Reiter, J.F. A cilium is not a cilium is not a cilium: signaling contributes to ciliary morphological diversity. Dev. Cell 14, 635–636 (2008).
Scholey, J.M. & Anderson, K.V. Intraflagellar transport and cilium-based signaling. Cell 125, 439–442 (2006).
Tissir, F. et al. Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus. Nat. Neurosci. 13, 700–707 (2010).
Han, Y.G. et al. Dual and opposing roles of primary cilia in medulloblastoma development. Nat. Med. 15, 1062–1065 (2009).
Wong, S.Y. et al. Primary cilia can both mediate and suppress Hedgehog pathway–dependent tumorigenesis. Nat. Med. 15, 1055–1061 (2009).
Han, Y.G. & Alvarez-Buylla, A. Role of primary cilia in brain development and cancer. Curr. Opin. Neurobiol. 20, 58–67 (2010).
Wilson, S.L., Wilson, J.P., Wang, C., Wang, B. & McConnell, S.K. Primary cilia and Gli3 activity regulate cerebral cortical size. Dev. Neurobiol. 72, 1196–1212 (2012).
Breunig, J.J. et al. Primary cilia regulate hippocampal neurogenesis by mediating sonic hedgehog signaling. Proc. Natl. Acad. Sci. USA 105, 13127–13132 (2008).
Amador-Arjona, A. et al. Primary cilia regulate proliferation of amplifying progenitors in adult hippocampus: implications for learning and memory. J. Neurosci. 31, 9933–9944 (2011).
Willaredt, M.A. et al. A crucial role for primary cilia in cortical morphogenesis. J. Neurosci. 28, 12887–12900 (2008).
Caspary, T., Larkins, C.E. & Anderson, K.V. The graded response to Sonic Hedgehog depends on cilia architecture. Dev. Cell 12, 767–778 (2007).
Hébert, J.M. & McConnell, S.K. Targeting of cre to the Foxg1 (BF-1) locus mediates loxP recombination in the telencephalon and other developing head structures. Dev. Biol. 222, 296–306 (2000).
Tronche, F. et al. Disruption of the glucocorticoid receptor gene in the nervous system results in reduced anxiety. Nat. Genet. 23, 99–103 (1999).
Zhuo, L. et al. hGFAP-cre transgenic mice for manipulation of glial and neuronal function in vivo. Genesis 31, 85–94 (2001).
Su, C.-Y., Bay, S.N., Mariani, L.E., Hillman, M.J. & Caspary, T. Temporal deletion of Arl13b reveals that a mispatterned neural tube corrects cell fate over time. Development 139, 4062–4071 (2012).
Mukhopadhyay, S., Lu, Y., Shaham, S. & Sengupta, P. Sensory signaling–dependent remodeling of olfactory cilia architecture in C. elegans. Dev. Cell 14, 762–774 (2008).
Kim, J. et al. Functional genomic screen for modulators of ciliogenesis and cilium length. Nature 464, 1048–1051 (2010).
Lehtinen, M.K. et al. The cerebrospinal fluid provides a proliferative niche for neural progenitor cells. Neuron 69, 893–905 (2011).
Zhu, D., Shi, S., Wang, H. & Liao, K. Growth arrest induces primary-cilium formation and sensitizes IGF-1–receptor signaling during differentiation induction of 3T3–L1 preadipocytes. J. Cell Sci. 122, 2760–2768 (2009).
Christensen, S.T., Clement, C.A., Satir, P. & Pedersen, L.B. Primary cilia and coordination of receptor tyrosine kinase signaling. J. Pathol. 226, 172–184 (2012).
Higginbotham, H. et al. Arl13b in primary cilia regulates the migration and placement of interneurons in the developing cerebral cortex. Dev. Cell 23, 925–938 (2012).
Sawamoto, K. et al. New neurons follow the flow of cerebrospinal fluid in the adult brain. Science 311, 629–632 (2006).
Cevik, S. et al. Joubert syndrome Arl13b functions at ciliary membranes and stabilizes protein transport in Caenorhabditis elegans. J. Cell Biol. 188, 953–969 (2010).
Horner, V.L. & Caspary, T. Disrupted dorsal neural tube BMP signaling in the cilia mutant Arl13b hnn stems from abnormal Shh signaling. Dev. Biol. 355, 43–54 (2011).
Wilsch-Bräuninger, M., Peters, J., Paridaen, J.T. & Huttner, W.B. Basolateral rather than apical primary cilia on neuroepithelial cells committed to delamination. Development 139, 95–105 (2012).
Sahara, S. & O'Leary, D.D. Fgf10 regulates transition period of cortical stem cell differentiation to radial glia controlling generation of neurons and basal progenitors. Neuron 63, 48–62 (2009).
Siegenthaler, J.A. & Pleasure, S.J. We have got you 'covered': how the meninges control brain development. Curr. Opin. Genet. Dev. 21, 249–255 (2011).
Brancati, F., Dallapiccola, B. & Valente, E.M. Joubert syndrome and related disorders. Orphanet J. Rare Dis. 5, 20 (2010).
Yokota, Y., Ring, C., Cheung, R., Pevny, L. & Anton, E.S. Nap1-regulated neuronal cytoskeletal dynamics is essential for the final differentiation of neurons in cerebral cortex. Neuron 54, 429–445 (2007).
Schmid, R.S. et al. Neuregulin 1-erbB2 signaling is required for the establishment of radial glia and their transformation into astrocytes in cerebral cortex. Proc. Natl. Acad. Sci. USA 100, 4251–4256 (2003).
Yokota, Y. et al. The adenomatous polyposis coli protein is an essential regulator of radial glial polarity and construction of the cerebral cortex. Neuron 61, 42–56 (2009).
Aruga, J., Nozaki, Y., Hatayama, M., Odaka, Y.S. & Yokota, N. Expression of ZIC family genes in meningiomas and other brain tumors. BMC Cancer 10, 79 (2010).
Berbari, N.F., Johnson, A.D., Lewis, J.S., Askwith, C.C. & Mykytyn, K. Identification of ciliary localization sequences within the third intracellular loop of G protein–coupled receptors. Mol. Biol. Cell 19, 1540–1547 (2008).
Acknowledgements
We thank A.-S. Lamantia, L. Pevny, M. Deshmukh and W. Snider for helpful comments and C.T. Strauss for editing. This research was supported by US National Institutes of Health grants MH060929 to E.S.A. and NS056380 to T.C., a NARSAD Young Investigator Award to H.H., a US National Institutes of Health predoctoral training grant to N.L.U. (T32GM008490) and the confocal imaging core of a National Institute of Neurological Disorders and Stroke institutional center core grant (P30 NS045892).
Author information
Authors and Affiliations
Contributions
E.S.A., H.H., J.G., Y.Y. and T.C. designed the experiments and supervised the project. H.H., J.G., Y.Y., N.L.U., C.-Y.S., J.L., V.G., J.H. and N.V. conducted the experiments and analyzed the data. E.S.A., H.H., J.G. and T.C. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures and Text
Supplementary Figures 1–5 (PDF 15089 kb)
Supplementary Video 1
MRI scan (axial plane) through E12.5 wild-type embryo showing normal CNS organization. (MOV 9651 kb)
Supplementary Video 2
MRI scan (axial plane) through E12.5 mutant embryo showing the perturbance in telencephalic CNS structures in mutants. (MOV 7455 kb)
Supplementary Video 3
Primary cilia dynamics in the ventricular zone. (AVI 7122 kb)
Supplementary Video 4
Primary cilium changes its localization in progenitors. (AVI 2865 kb)
Supplementary Video 5
Wild-type primary cilia dynamics. (AVI 914 kb)
Supplementary Video 6
Arl13b mutant primary cilia dynamics. (AVI 772 kb)
Rights and permissions
About this article
Cite this article
Higginbotham, H., Guo, J., Yokota, Y. et al. Arl13b-regulated cilia activities are essential for polarized radial glial scaffold formation. Nat Neurosci 16, 1000–1007 (2013). https://doi.org/10.1038/nn.3451
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3451
This article is cited by
-
How mechanisms of stem cell polarity shape the human cerebral cortex
Nature Reviews Neuroscience (2022)
-
Modelling ciliopathy phenotypes in human tissues derived from pluripotent stem cells with genetically ablated cilia
Nature Biomedical Engineering (2022)
-
Arl13b controls basal cell stemness properties and Hedgehog signaling in the mouse epididymis
Cellular and Molecular Life Sciences (2022)
-
The localization of amyloid precursor protein to ependymal cilia in vertebrates and its role in ciliogenesis and brain development in zebrafish
Scientific Reports (2021)
-
Induction of inverted morphology in brain organoids by vertical-mixing bioreactors
Communications Biology (2021)