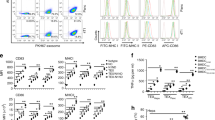
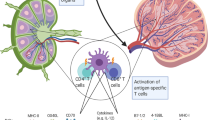
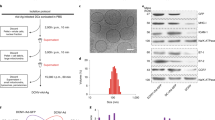
Abstract
Dendritic cells (DCs) are professional antigen presenting cells with the unique capacity to induce primary and secondary immune responses in vivo. Here, we show that DCs secrete antigen presenting vesicles, called exosomes, which express functional Major Histocompatibility Complex class I and class II, and T-cell costimulatory molecules. Tumor peptide-pulsed DC-derived exosomes prime specific cytotoxic T lymphocytes in vivo and eradicate or suppress growth of established murine tumors in a T cell-dependent manner. Exosome-based cell-free vaccines represent an alternative to DC adoptive therapy for suppressing tumor growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Boon, T. & van der Bruggen, P. Human tumor antigens recognized by T lymphocytes. J. Exp. Med 183, 725–729 (1996).
Lotze, M.T. et al. Cytokine gene therapy of cancer using 1L-12: murine and clinical trials. Ann. NY Acad. Sci. 795, 440–454 (1996).
Restifo, N. The new vaccines: building viruses that elicit antitumor immunity. Curr. Opin. Immunol. 8, 658–663 (1996)
Tüting, T., DeLeo, A.B., Lotze, M. & Storkus, w. Genetically modified bone marrow-derived dendritic cells expressing tumor-associated viral or “self” antigens induce antitumor immunity in vivo. Eur. J. Immunol. 27, 2702–2707 (1997).
Cayeux, S. Influence of gene-modified (IL-7, IL-4, and B7) tumor cell vaccines on tumor antigen presentation. J. Immunol. 158, 2834–2841 (1997).
Iwasaki, A. et al. The dominant role of bone marrow-derived cells in CTL induction following plasmid DNA immunization at different sites. J. Immunol. 159, 11–14 (1997).
Hart, D.N.J. Dendritic cells: unique leukocyte populations which control the primary immune responses. Blood 90, 3245–3287 (1997).
Caux, C., Dezutter-Dambuyant, C., Schmitt, D. & Banchereau, J. GM-CSF and TNFα cooperate in the generation of dendritic Langherans cells. Nature 360, 258 (1992).
Pope, M., Betjes, M., Hirmand, H., Hoffman, L. & Steinman, R. Both dendritic cells and memory T lymphocytes emigrate from organ cultures of human skin and form distinctive dendritic-T cell conjugates. J. Invest. Dermatol. 104, 11 (1995).
Inaba, K., Metlay, J., Crowley, M. & Steinman, R. Dendritic cells pulsed with protein antigens in vitro can prime antigen-specific, MHC-restricted T cell responses in situ. J. Exp. Med. 172, 631–640 (1990).
Steinman, R.M. The dendritic cell system and its role in immunogenicity. Annu. Rev. Immunol. 9, 271–296 (1991).
Ambe, K., Mori, M. & Enjoji, M. S100 protein positive dendritic cells in colorectal adenocarcinomas. Distribution and relation to the clinical prognosis. Cancer 63, 496 (1989).
Maraskovsky, E. et al. Dramatic increase in the number of functionally mature dendritic cells in Flt3 ligand-treated mice: multiple dendritic cell subpopulations identified. J Exp. Med. 184, 1953–1962 (1996).
Girolomoni, C. & Ricciardi-Castagnoli, P. Dendritic cells hold promise for im-munotherapy. Immunol. Today 18, 102–104 (1997).
Germain, R.N. MHC-dependent antigen processing and peptide presentation: providing ligands for T lymphocyte activation. Cell 76, 287–299 (1994).
Pierre, P. et al. Developmental regulation of MHC class II transport in mouse dendritic cells. Nature 388, 787–792 (1997).
Cella, M., Engering, A., Pinet, V., Pieters, J. & Lanzavecchia, A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 388, 782–786 (1997).
Kleijmeer, M.,J., Morkowski, S., Griffith, J., M., Rudensky, A., Y. & Geuze, H., J. MHC class II compartments in human and mouse B lymphoblasts represent conventional endocytic compartments. J. Cell. Biol. 139, 639–649 (1997).
Raposo, G. et al. B lymphocytes secrete antigen presenting vesicles. J. Exp. Med. 183, 1161–1172 (1996).
Dufour, E. et al. Diversity of the cytotoxic melanoma-specific immune response. J. Immunol. 158, 3787–3795 (1997).
Mayordomo, J.I. et al. Bone marrow-derived dendritic cells pulsed with synthetic tumour peptides elicit protective and therapeutic antitumour immunity. Nature Med. 1, 1297–1302 (1995).
Winzler, C. et al. Maturation stages of mouse dendritic cells in growth factor-dependent long term cultures. J. Exp. Med. 185, 317–328 (1997).
Zitvogel, L. et al. Therapy of murine tumors with tumor peptide-pulsed dendritic cells: dependence on T cells, B7 costimulation, and T helper cell 1-associated cytokines. J. Exp. Med. 183, 87–97 (1996).
Warnier, G. et al. Induction of a cytolytic T-cell response in mice with a recombinant adenovirus coding for tumor antigen P815A. Int. J. Cancer 67, 303–310 (1996).
Zitvogel, L. et al. B7.1 costimulation markedly enhances IL-12 mediated antitumor immunity in vivo. Eur. J. Immunol. 26, 1335–1341 (1996).
Gabrilovich, D.I., Ciernik, F. & Carbone, D.,P. Dendritic cells in antitumor immune responses: defective antigen presentation in tumor-bearing hosts. Cell. Immunol. 170,101 (1996).
Bender, A., Sapp, M., Schuler, G., Steinman, R.M. & Bhardwaj, N. Improved methods for the generation of dendritic cells from nonproliferating progenitors in human blood. J. Immunol. Methods. 196, 121–135 (1996).
Schmidt, W. et al. Cell-free tumor antigen peptide-based cancer vaccines. Proc. Natl. Acad. Sci. USA 94, 3262–3267 (1997).
Raposo, G., Kleijmeer, M., Posthuma, J., Slot, G. & Geuze, H. in Handbook of Exp. Immunol. 5th ed. (eds Herzenberg, L.A., Weir, D.M., Herzenberg, L.A. & Blackwell, C.) 1–11 (Science Inc. Maiden MA., 1997).
Sallusto, F. & Lanzavecchia, A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor a. J. Exp. Med. 179, 1109–1118 (1994).
Bockxmeer, F.v. & Morgan, E. Transferrin receptors during rabbit reticulocyte maturation. Biochim. Biophys. Acta 584, 76–83 (1979).
Stam, N., Spits, H. & Ploegh, H. Monoclonal antibodies raised against denaturated HLA-A and HLA-B locus H-chain permit biochemical characterization of certain HLA-C locus products. J. Immunol. 137, 2299–2306 (1986).
Metzelaar, M. et al. CD63 antigen: a novel lysosomal membrane glycoprotein, cloned by a screening procedure for intracellular antigens in eukaryotic cells. J. Biol. Chem. 266, 3239–3245 (1991).
Mayordomo, J.I. et al. Bone-marrow derived DC serve as potent adjuvants for peptide-based antitumor vaccines. Stem Cells 15, 94–103 (1997).
Caux, C. et al. B70/B7.2 is identical to CD86 and is the major functional ligand for CD28 expressed on human dendritic cell. J. Exp. Med. 180, 1841–1847 (1994)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zitvogel, L., Regnault, A., Lozier, A. et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell derived exosomes. Nat Med 4, 594–600 (1998). https://doi.org/10.1038/nm0598-594
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0598-594
This article is cited by
-
Modification of immune cell-derived exosomes for enhanced cancer immunotherapy: current advances and therapeutic applications
Experimental & Molecular Medicine (2024)
-
Identification of an exosome-related signature associated with prognosis and immune infiltration in breast cancer
Scientific Reports (2023)
-
Description and optimization of a multiplex bead-based flow cytometry method (MBFCM) to characterize extracellular vesicles in serum samples from patients with hematological malignancies
Cancer Gene Therapy (2022)
-
Extracellular vesicles: from bench to bedside
Current Medicine (2022)
-
Perspectives and Challenges on the Potential Use of Exosomes in Bioartificial Pancreas Engineering
Annals of Biomedical Engineering (2022)