Abstract
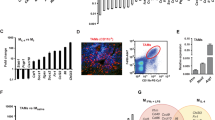
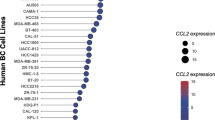
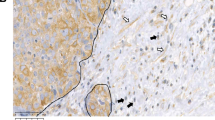
Macrophages, which are abundant in the tumour microenvironment, enhance malignancy1. At metastatic sites, a distinct population of metastasis-associated macrophages promotes the extravasation, seeding and persistent growth of tumour cells2. Here we define the origin of these macrophages by showing that Gr1-positive inflammatory monocytes are preferentially recruited to pulmonary metastases but not to primary mammary tumours in mice. This process also occurs for human inflammatory monocytes in pulmonary metastases of human breast cancer cells. The recruitment of these inflammatory monocytes, which express CCR2 (the receptor for chemokine CCL2), as well as the subsequent recruitment of metastasis-associated macrophages and their interaction with metastasizing tumour cells, is dependent on CCL2 synthesized by both the tumour and the stroma. Inhibition of CCL2–CCR2 signalling blocks the recruitment of inflammatory monocytes, inhibits metastasis in vivo and prolongs the survival of tumour-bearing mice. Depletion of tumour-cell-derived CCL2 also inhibits metastatic seeding. Inflammatory monocytes promote the extravasation of tumour cells in a process that requires monocyte-derived vascular endothelial growth factor. CCL2 expression and macrophage infiltration are correlated with poor prognosis and metastatic disease in human breast cancer3,4,5,6. Our data provide the mechanistic link between these two clinical associations and indicate new therapeutic targets for treating metastatic breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Qian, B. Z. & Pollard, J. W. Macrophage diversity enhances tumor progression and metastasis. Cell. 141, 39–51 (2010)
Qian, B. et al. A distinct macrophage population mediates metastatic breast cancer cell extravasation, establishment and growth. PLoS ONE 4, e6562 (2009)
Ueno, T. et al. Significance of macrophage chemoattractant protein-1 in macrophage recruitment, angiogenesis, and survival in human breast cancer. Clin. Cancer Res. 6, 3282–3289 (2000)
Valkovic, T., Lucin, K., Krstulja, M., Dobi-Babic, R. & Jonjic, N. Expression of monocyte chemotactic protein-1 in human invasive ductal breast cancer. Pathol. Res. Pract. 194, 335–340 (1998)
Saji, H. et al. Significant correlation of monocyte chemoattractant protein-1 expression with neovascularization and progression of breast carcinoma. Cancer 92, 1085–1091 (2001)
Rhodes, D. R. et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6, 1–6 (2004)
Geissmann, F. et al. Blood monocytes: distinct subsets, how they relate to dendritic cells, and their possible roles in the regulation of T-cell responses. Immunol. Cell Biol. 86, 398–408 (2008)
Geissmann, F. et al. Development of monocytes, macrophages, and dendritic cells. Science 327, 656–661 (2010)
Geissmann, F., Jung, S. & Littman, D. R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003)
Getts, D. R. et al. Ly6c+ “inflammatory monocytes” are microglial precursors recruited in a pathogenic manner in West Nile virus encephalitis. J. Exp. Med. 205, 2319–2337 (2008)
Palframan, R. T. et al. Inflammatory chemokine transport and presentation in HEV: a remote control mechanism for monocyte recruitment to lymph nodes in inflamed tissues. J. Exp. Med. 194, 1361–1374 (2001)
Borowsky, A. D. et al. Syngeneic mouse mammary carcinoma cell lines: two closely related cell lines with divergent metastatic behavior. Clin. Exp. Metastasis 22, 47–59 (2005)
Tsui, P. et al. Generation, characterization and biological activity of CCL2 (MCP-1/JE) and CCL12 (MCP-5) specific antibodies. Hum. Antibodies 16, 117–125 (2007)
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005)
Carton, J. M. et al. Codon engineering for improved antibody expression in mammalian cells. Protein Expr. Purif. 55, 279–286 (2007)
Al-Mehdi, A. B. et al. Intravascular origin of metastasis from the proliferation of endothelium-attached tumor cells: a new model for metastasis. Nature Med. 6, 100–102 (2000)
Ma, C. & Wang, X.-F. In vitro assays for the extracellular matrix protein-regulated extravasation process. CSH Protoc 10.1101/pdb.prot5034 (2008)
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009)
Gerber, H. P. et al. VEGF is required for growth and survival in neonatal mice. Development 126, 1149–1159 (1999)
Huang, Y. et al. Pulmonary vascular destabilization in the premetastatic phase facilitates lung metastasis. Cancer Res. 69, 7529–7537 (2009)
Nielsen, B. S. et al. A precise and efficient stereological method for determining murine lung metastasis volumes. Am. J. Pathol. 158, 1997–2003 (2001)
Abramoff, M. D., Magelhaes, P. J. & Ram, S. J. Image processing with ImageJ. Biophotonics Int. 11, 36–42 (2004)
Havens, A. M. et al. An in vivo mouse model for human prostate cancer metastasis. Neoplasia 10, 371–380 (2008)
Brandt, B. et al. 3D-extravasation model — selection of highly motile and metastatic cancer cells. Semin. Cancer Biol. 15, 387–395 (2005)
Stern, P. et al. A system for Cre-regulated RNA interference in vivo . Proc. Natl Acad. Sci. USA 105, 13895–13900 (2008)
Im, J. H. et al. Coagulation facilitates tumor cell spreading in the pulmonary vasculature during early metastatic colony formation. Cancer Res. 64, 8613–8619 (2004)
Acknowledgements
This work was supported by grants from the NIH to J.W.P. (NIH PO1 CA100324 and RO1 CA131270) and to the Albert Einstein Cancer Center Core (P30 CA 13330). We thank J. Massague for 4173 cells and N. Ferrara for the Vegfaflox/flox mice. We also thank P. Marsters for statistical analyses and M. Thompson, F. Shi, C. Ferrante, F. McCabe, H. Millar-Quinn and D. Wiley for discussions and technical assistance.
Author information
Authors and Affiliations
Contributions
B-Z.Q., L.A.S. and J.W.P. conceived the ideas and designed the experiments. B-Z.Q., J.L., H.Z., T.K., J.Z., L.R.C. and E.A.K. performed the experiments. B-Z.Q., J.L., L.A.S. and J.W.P. analysed the data. B-Z.Q., L.A.S. and J.W.P. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-11 with legends, Supplementary Movie legends and additional references. (PDF 703 kb)
Supplementary Movie 1
The movie shows representative 3D reconstructed confocal images of tumor cells (CFP, blue) and macrophages (GFP, green) 24 hours after tumor cell tail veininjection in mice treated with control Ab (see Supplementary Information file for full legend). (AVI 2244 kb)
Supplementary Movie 2
The movie shows representative 3D reconstructed confocal images of tumor cells (CFP, blue) and macrophages (GFP, green) 24 hours after tumor cell tail vein injection in mice treated with anti-mouse CCL2 Ab (see Supplementary Information file for full legend). (AVI 1529 kb)
Rights and permissions
About this article
Cite this article
Qian, BZ., Li, J., Zhang, H. et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475, 222–225 (2011). https://doi.org/10.1038/nature10138
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10138
This article is cited by
-
Cell-cell communication characteristics in breast cancer metastasis
Cell Communication and Signaling (2024)
-
Cell fusion upregulates PD-L1 expression for evasion from immunosurveillance
Cancer Gene Therapy (2024)
-
How chemokines organize the tumour microenvironment
Nature Reviews Cancer (2024)
-
Crosstalk between endothelial progenitor cells and HCC through periostin/CCL2/CD36 supports formation of the pro-metastatic microenvironment in HCC
Oncogene (2024)
-
Neutrophils in cancer: dual roles through intercellular interactions
Oncogene (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.