Abstract
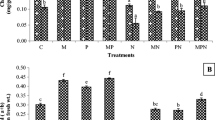
The effects of Glomus mosseae and Paecilomyces lilacinus on Meloidogyne javanica of tomato were tested in a greenhouse experiment. Chicken layer manure was used as a carrier substrate for the inoculum of P. lilacinus. The following parameters were used: gall index, average number of galls per root system, plant height, shoot and root weights. Inoculation of tomato plants with G. mosseae did not markedly increase the growth of infected plants with M. javanica. Inoculation of plants with G. mosseae and P. lilacinus together or separately resulted in similar shoots and plant heights. The highest root development was achieved when mycorrhizal plants were inoculated with P. lilacinus to control root-knot nematode. Inoculation of tomato plants with G. mosseae suppressed gall index and the average number of galls per root system by 52% and 66%, respectively, compared with seedlings inoculated with M. javanica alone. Biological control with both G. mosseae and P. lilacinus together or separately in the presence of layer manure completely inhibited root infection with M. javanica. Mycorrhizal colonization was not affected by the layer manure treatment or by root inoculation with P. lilacinus. Addition of layer manure had a beneficial effect on plant growth and reduced M. javanica infection.
Similar content being viewed by others
References
Abu-Gharbieh W (1988) Root-knot nematodes: Meloidogyne spp. in Jordan. Biology and control (in Arabic). University of Jordan, Amman
Abu-Laban A (1991) Evaluation of three nematophagous fungi in controlling root-knot nematode using animal manure and wheat grain as carrier substrates. MSc thesis, University of Jordan
Al-Raddad A (1987) Effect of three vesicular-arbuscular mycorrhizal isolates on growth of tomato, eggplant and pepper in a field soil. Dirasat 14:161–168
Atilano R, Menge J, Van Gundy S (1981) Interaction between Meloidogyne arenaria and Glomus fasciculatum in grape. J Nematol 13:52–57
Barker K (1985) Nematode extraction and bioassays. In: Barker K, Carter C, Sasser J (eds) An advanced treatise on Meloidogyne, vol II. North Carolina State University Graphics, Raleigh
Biermann B, Lindermann R (1981) Quantifying vesicular-arbuscular mycorrhizae: a proposed method towards standardization. New Phytol 87:63–67
Cabanillas E, Barker K (1989) Impact of Paecilomyces lilacinus inoculum level and application time on control of Meloidogyne incognita on tomato. J Nematol 21:115–120
Cooper K, Grandison G (1986) Interaction of vesicular-arbuscular mycorrhizal fungi with root-knot nematode on cultivars of tomato and white clover susceptible to Meloidogyne hapla. Ann Appl Biol 108:1–11
Dehne H (1982) Interaction between vesicular-arbuscular mycorrhizal fungi and plant pathogens. Phytopathology 72:1115–1119
Francl L, Dropkin V (1985) Glomus fasciculatum, a weak pathogen of Heterodera glycines. J Nematol 17:470–475
Grandison G, Cooper K (1986) Interaction of vesicular-arbuscular mycorrhizae and cultivars of alfalfa susceptible and resistant to Meloidogyne hapla. J Nematol 18:141–149
Hussey R, Roncadori R (1977) Interaction of Pratylenchus brachyurus and Gigaspora margarita endomycorrhizal fungus on cotton. J Nematol 10:16–20
Hussey R, Roncadori R (1982) Vesicular-arbuscular mycorrhizae may limit nematode activity and improve plant growth. Plant Dis 66:9–14
Jatala P (1986) Biological control of plant parasitic nematodes. Annu Rev Phytopathol 24:453–489
Jenkins W, Taylor D (1967) Plant nematology. Reinhold, New York
Menge J, Timmer L (1982) Procedures for inoculation of plants with vesicular-arbuscular mycorrhizae in the laboratory, greenhouse and field. In: Schenck N (ed) Methods and principles of mycorrhizal research. American Phytopathology Society, St Paul, Minn
Phillips J, Hayman D (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid movement of infection. Trans Br Mycol Soc 55:158–161
Qadri A (1989) Fungi associated with the sugar beet cyst nematode Heterodera schatii in Jerash, Jordan. MSc thesis, University of Jordan
Rodriguez-Kabana R, Morgan-Jones G, Chet I (1987) Biological control of nematodes: soil amendments and microbial activity. Plant Soil 100:237–247
Roncadori R, Hussey R (1977) Interaction of the endomycorrhizal fungus Gigaspora margarita and root-knot nematode on cotton. Phytopathology 67:1507–1511
Saleh H, Sikora R (1984) Relationship between Glomus fasciculatum root colonization and its effect on Meloidogyne incognita. Nematologica 30:230–237
Sikora R (1978) Einfluss der endotrophen Mykorrhiza (Glomus mosseae) auf das Wirt-Parasit Verhältnis von Meloidogyne incognita in Tomaten. Z Pflanzenkr Pflanzensch 85:197–202
Southey J (1970) Laboratory methods for work with plant and soil nematodes, 5th edn. (Fish and food technology bulletin 2) Ministry of Agriculture, London
Van Gundy SD, Kirkpatrick JD (1975) Nematode-nematode interactions on tomato. J Nematol 7:330–331
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Al-Raddad, A.M. Interaction of Glomus mosseae and Paecilomyces lilacinus on Meloidogyne javanica of tomato. Mycorrhiza 5, 233–236 (1995). https://doi.org/10.1007/BF00203344
Issue Date:
DOI: https://doi.org/10.1007/BF00203344