Summary
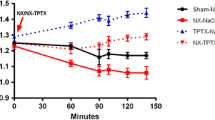
The release of cyclic AMP from bone in response to stimulation with PTH 1–34 was examined in 20 dogs with long-term chronic renal failure (CRF) produced by unilateral nephrectomy and contralateral partial renal artery ligation. After 9 to 15 months of uremia, the tibiae were removed and perfused in vitro. Seven dogs with CRF served as controls, 7 dogs with CRF were treated with 24,25(OH)2D3 — 2.5 µg per day, and 6 CRF dogs underwent thyroparathyroidectomy (TPTX) 42 h before they were sacrificed. The release of cyclic AMP from bone in response to PTH 1–34 in the CRF dogs was severely reduced compared to the response observed in 7 dogs with normal renal function (net accumulation of cyclic AMP release 86±8.5 versus 426±59.0 pmol/30 min). Long-term treatment of uremic dogs with 24,25(OH)2D3 had no effect on the release of cyclic AMP by bone. However, the release of cyclic AMP was restored to normal levels in the CRF dogs that underwent thyroparathyroidectomy. All CRF dogs had secondary hyperparathyroidism and the fact that TPTX returned the cyclic AMP response to normal values suggests that desensitization to PTH of the adenylate cyclase system of bone exists in chronic uremia.
Similar content being viewed by others
References
Evanson JM (1966) The response to the infusion of parathyroid extract in hypocalcemic states. Clin Sci 31:63–75
Massry SG, Coburn JW, Lee DBN, Jowsey J, Kleeman CR (1973) Skeletal resistance to parathyroid hormone in renal failure. Ann Intern Med 78:357–364
Llach F, Massry SG, Singer FR, Kurokawa K, Kaye JH, Coburn JW (1975) Skeletal resistance to endogenous parathyroid hormone in patients with early renal failure. A possible cause for secondary hyperparathyroidism. J Clin Endocrinol Metab 41:339–345
Massry SG, Stein R, Garty J, Arieff AL, Coburn JW, Norman AW, Friedler RM (1976) Skeletal resistance to the calcemic action of parathyroid hormone in uremia: Role of 1,25(OH)2D3. Kidney Int 9:467–474
Somerville PJ, Kaye M (1978) Resistance to parathyroid hormone in renal failure: Role of vitamin D metabolites. Kidney Int 14:245–254
Somerville PJ, Kaye M (1979) Evidence that resistance to the calcemic action of parathyroid hormone in rats with acute uremia is caused by phosphate retention. Kidney Int 16:552–560
Olgaard K, Finco D, Martin K, Schwartz J, Korkor A, Bellorin-Font E, Klahr S, Slatopolsky E (1981) The extraction of parathyroid hormone and the generation of cyclic AMP by isolated perfused bones from acutely uremic dogs. Abstract, VII Parathyroid Conference. In: Cohn DV, Talmage RV, Matthews JL (eds) Hormonal Control of Calcium Metabolism. Excerpta Medica, Princeton, p 190
Martin KJ, Freitag JJ, Conrades MB, Hruska KA, Klahr S, Slatopolsky E (1978) Selective uptake of the synthetic amino terminal fragment of bovine parathyroid hormone by isolated perfused bone. J Clin Invest 62:856–861
Hruska KA, Kopelman R, Rutherford WE, Klahr S, Slatopolsky E (1975) Metabolism of immunoreactive parathyroid hormone in the dog. The role of the kidney and the effects of chronic renal disease. J Clin Invest 56:39–48
Steiner AL, Kipnis DM, Utiger R, Parker C (1969) Radioimmunoassay for the measrement of adenosine 3′,5′-cyclic phosphate. Proc Natl Acad Sci USA 64:367–373
Jacob AI, Gavellas G, Canterbury JM, Bourgoignie JJ (1981) Normal calcemic response to bPTH (1–34) but not bPTH (1–84) in uremia. Abstract CA-077, The Int Meeting of Nephrology, Greece
Hruska KA, Korkor A, Martin K, Slatopolsky E (1981) Peripheral metabolism of intact parathyroid hormone. Role of liver and kidney and the effect of chronic renal failure. J Clin Invest 67:885–892
Marcus R, Orner FB, Brickman AS (1980) Effects in vivo of vitamin D metabolites and 17-estradiol on parathyroid hormone-dependent formation of adenosine 3′,5′-monophosphate in rat bone. Endocrinology 107:1593–1599
Peacock M, Taylor GA, Redel J (1976) The action of two metabolites of vitamin D3; 25,26-dihydroxycholecalciferol (25,26(OH)2D3) and 24,25-dihydroxycholecalciferol (24,25-(OH)2D3) on bone resorption. FEBS Lett 62:248–250
Queille ML, Miravet L, Bordier P, Redel J (1978) The action of vitamin D metabolites (25(OH)D3-1,25(OH)2D3-24,25-(OH)2D3-25,26(OH)2D3) on vitamin D deficient rats. Biomedicine 28:237–242
Lieberherr M, Garabedian M, Guillozo H, Bailly du Bois M, Balsan S (1979) Interaction of 24,25-dihydroxyvitamin D3 and parathyroid hormone on bone emzymes in vitro. Calcif Tissue Int 27:47–52
Lieberherr M, Garabedian M, Guillozo H, Thil CL, Balsan S (1980) In vitro effects of vitamin D3 metabolites on rat calvaria cAMP content. Calcif Tissue Int 30:209–216.
Tanaka Y, DeLuca HF, Kobayashi Y, Taguchi T, Ikekawa N, Morisaki M (1979) Biological activity of 24,24-difluoro-25-hydroxyvitamin D3: Effect of blocking of 24-hydroxylation on the functions of vitamin D. J Biol Chem 254:7163–7167
Llach F, Brickman AS, Singer FR, Coburn JW (1979) 24,25-dihydroxycholecalciferol, a vitamin D sterol with qualitatively unique effects in uremic man. Metab Bone Dis & Rel Res 2:11–15
Catt KJ, Harwood JP, Aguilera G, Dufau ML (1979) Hormonal regulation of peptide receptors and target cell responses. Nature 280:109–116
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Olgaard, K., Arbelaez, M., Schwartz, J. et al. Abnormal skeletal response to parathyroid hormone in dogs with chronic uremia. Calcif Tissue Int 34, 403–407 (1982). https://doi.org/10.1007/BF02411275
Issue Date:
DOI: https://doi.org/10.1007/BF02411275