Summary
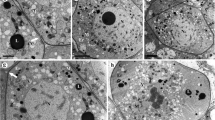
Ultrastructural studies made on the micropyle of sunflower before and after pollination resulted in the following observations. (1) The micropyle is closed instead of a hole or canal. The inner epidermis of the integument on both sides of the micropyle is in close contact at the apex of the ovule. The boundary between the two sides consists of two layers of epidermal cuticle. (2) The micropyle contains a transmitting tissue. The micropyle is composed of an intercellular matrix produced by the epidermal cells of the integument. (3) The micropyle is asymmetrical, and is much wider on the side proximal to the funicle. On the funicle side the cells adjacent to the micropyle are similar to those of the transmitting tissue: they have large amounts of intercellular matrix and contain abundant dictyosomes, rough ER, and starch grains, and provide an appropriate environment for growth of the pollen tubes. The cells distal to the funicle are rich in rough ER and lipid bodies; they lack large intercellular spaces. (4) The micropyle is variable in the axial direction, i.e., it is much larger and more asymmetric at the level distal to the embryo sac than at a level close to the embryo sac. After pollination, one to four pollen tubes are seen in a micropyle. During their passage through the micropyle, most pollen tubes are restricted to the side proximal to the funicle. There is a greater tendency (81%) for the degenerate synergid to be located toward the funicle, i.e., at the same side as the pollen tube pathway. The data indicate a close relationship between micropyle organization, orientation of pollen tube growth, and synergid degeneration.
Similar content being viewed by others
References
Bell J, Hicks G (1976) Transmitting tissue in the pistil of tobacco: light and electron microscopic observations. Planta 131:187–200
Brough P (1933) The life history of Grevillea robusta Cunn. Proc Linn Soc N S Wales 58:33–73
Chao CY (1971) A periodic acid-Schiff's substance related to the directional growth of pollen tube into embryo sac in Paspalum ovules. Am J Bot 58:649–654
Chaubal R, Reger B (1990) Relatively high calcium is localized in synergid cells of wheat ovaries. Sex Plant Reprod 3:98–102
Ciampolini F, Cresti M, Pacini E (1978) Carateeristiche ultrastructturali ed istochimiche del tessuto trasmittente stilare di melo. In: Proc Symp “Gertilita delle piante da frutto”. Bologna, pp 544–551
Condit IJ (1932) The structure and development of flowers in Ficus carica. Hilgardia 6:443–484
Cooper DC (1938) Embryology of Pisum sativum. Bot Gaz 100:123–132
Cresti M, Van Went JL, Pacini E, Willemse MTM (1976) Ultrastructure of transmitting tissue of Lycopersicon Peruvianum style: Development and histochemistry. Planta 132:305–312
Cresti M, Ciampolini F, Pacini E (1978) Gamma irradiation of Prunus arium flower buds: effects on stylar development-An ultrastructural study. Acta Bot Neerl 27:97–106
Dashek WV, Thomas HR, Rosen WG (1971) Secretory cell of lily pistils. II. Electron microscope cytochemistry of canal cells. Am J Bot 58:909–920
Engels FM (1974) Function of Golgi vesicles in relation to cell wall synthesis in germinating petunia pollen II. Chemical composition of golgi vesicles and pollen tube wall. Acta Bot Neerl 23:81–89
Gelin OEV (1936) Zur Embryologie und Zytologie van Berkheya bergiana Soderb. du B. aldami Hook. Sven Bot Tidskr 30:324–328
Grootjen CJ, Bouman F (1981) Development of ovule and seed in Costus cuspidatus (Zingiberaceae) with special reference to the operculum. Bot J Linn Soc 83:29–39
Jensen WA, Fisher DB (1969) Cotton embryogenesis: The tissue of the stigma and style and their relation to pollen tube. Planta 84:97–121
Jensen WA, Fisher DB (1970) Cotton embryogenesis: The pollen tube in the stigma and style. Protoplasma 69:215–235
Jensen WA, Ashton ME, Beasley CA (1983) Pollen tube-embryo sac interaction in cotton. In: Mulcahy DL, Ottaviano E (eds) Pollen: biology and implications for plant breeding. Elsevier Sci Publ, Amsterdam, pp 67–72
Khan R (1943) The ovule and embryo sac of Fouguieria. Proc Natl Inst Sci India 9:253–256
Knox RB (1984) Pollen-pistil interaction. In: Linskens HF, HeslopHarrison J (eds) Cellular interactions. Springer, Berlin Heidelberg New York, pp 508–608
Kroh M (1973) Nature of the intercellular substance of stylar transmitting tissue. In: Loewus F (ed) Biogenesis of plant cell wall polysaccharides. Academic Press, New York, pp 195–205
Kroh M, Helsper JPFG (1974) Transmitting tissue and pollen tube growth. In: Linskens (ed) Fertilizations in higher plants. NorthHolland Publ, Amsterdam, pp 167–175
Kroh M, Van Bakel CHJ (1973) Incorporation of label into the intercellular substance of stylar transmitting tissue from Petunia pistils labelled with tritiated myo-inositol. An electron microscopic autoradiographic study. Acta Bot Neerl 22:106–111
Kroh M, Gorissen MH, Pfahler PL (1979) Ultrastructure studies on styles and pollen tubes of Zea mays L. general survey on pollen tube growth in vivo. Acta Bot Neerl 28:513–518
Linskens HF (1974a) Some observations on the growth of the style. Incomp Newsl 4:4–15
Linskens HF (1974b) Study of growth of Petunia styles. Fyziol Rast 21:1059–1064
Loewus F, Labarca C (1973) Pistil secretion product and pollen tube wall formations. In: Loewus F (ed) Biogenesis of plant cell wall polysaccharides. Academic Press, New York, pp 175–193
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGraw-Hill, New York Toronto London
Mascarenhas JP (1962a) Chemotropic response of Antirrhinum majus pollen to calcium. Nature 196:292–293
Mascarenhas JP, Machlis L (1962b) The pollen tube chemotropic factor from Antirrhinum majus: Bioassay, extraction, and partial purification. Am J Bot 49:482–489
Mascarenhas JP, Machlis L (1962c) The hormonal control of directional growth of pollen tubes. Vitam Horm 20:347–371
Pope MN (1946) The course of the pollen tube in cultivated barley. J Am Soc Agron 38:432–440
Stanley RG, Linskens HF (1974) Pollen. Biology biochemistry management. Springer, Berlin Heidelberg New York
Steindl F (1945) Beitrag Zur pollen- und embryobildung beri Cynomorium coccineum L. Arch Julius Klaus-Stift f Vererbungstorsch 20:342–355
Van Went JL, Willemse MTM (1984) Fertilization. In: Johri BM (ed) Embryology of angiosperms. Springer, Berlin Heidelberg New York Tokyo, pp 273–317
Vasil IK (1974) The histology and physiology of pollen germination and pollen tube growth on the stigma and in the style. In: Linskens HF (ed) Fertilization in higher plants. North-Holland Publ, Amsterdam, pp 105–117
Vithanage HIMV, Knox RB (1977) Development and cytochemistry of stigma surface and response to self- and foreign pollination in Helianthus annuus. Phytomorphology 27:168–179
Weinstein AJ (1926) Cytological studies on Phaseolus vulgaris. Am J Bot 13:248–263
Wilms HJ (1980a) Ultrastructure of the stigma and style of spinach in relation to pollen germination and pollen tube growth. Acta Bot Neerl 29:33–47
Wilms HJ (1980b) Development of composition of the spinach ovule. Acta Bot Neerl 29:243–260
Yan H, Yang HY, Jensen WA (1990a) Ultrastructure of sunflower embryo sac in respect to the concept of female germ unit. Acta Bot Sin 32:165–171
Yan H, Yang HY, Jensen WA (1990b) Ultrastructure of developing embryo sac of sunflower (Helianthus annuus) before and after fertilization. Can J Bot (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yan, H., Yang, Hy. & Jensen, W.A. Ultrastructure of the micropyle and its relationship to pollen tube growth and synergid degeneration in sunflower. Sexual Plant Reprod 4, 166–175 (1991). https://doi.org/10.1007/BF00190000
Issue Date:
DOI: https://doi.org/10.1007/BF00190000