Abstract
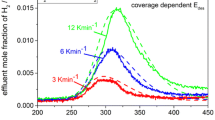
The kinetics of desorption of hydrogen from the copper component of an alumina-supported polycrystalline copper catalyst has been studied in detail by temperature-programmed desorption (TPD). Line-shape analysis of the hydrogen TPD spectra shows: (i) that the desorption is second order, (ii) that the desorption activation energy is in the range 64–68 kJ mol−1 in the coverage range 7–44% of a monolayer, and (iii) that the desorption pre-exponential term has a value ∼10−5 cm2 s−1 atom−1 consistent with the desorption being second order, involving mobile adsorbates and a mobile desorption transition state.
Similar content being viewed by others
References
W.X. Pan, R. Cao, D.L. Roberts and G.L. Griffin, J. Catal. 114 (1988) 440.
G.C. Chinchen, K.C. Waugh and D.A. Whan, Appl. Catal. 25 (1986) 101.
Yu.B. Kagan, L.G. Liberov, E.V. Slivinski, S.M. Lockev, G.I. Lin, A.Ya. Rozovsky and A.N. Bashirov, Dokl. Akad. Nauk SSSR 221 (1975) 1093.
G.C. Chinchen, P.J. Denny, D.G. Parker, G.D. Short, M.S. Spencer, K.C. Waugh and D.A. Whan, Prepr. Am. Chem. Soc. Div. Fuel Chem. 29 (1984) 178.
G.J. Millar, C.H. Rochester, C. Howe and K.C. Waugh, J. Mol. Phys. 76 (1991) 833.
M. Bowker, R.A. Hadden, H. Houghton, J.N.K. Hyland and K.C. Waugh, J. Catal. 109 (1988) 263.
S. Bailey, G.F. Froment, J.W. Snoeck and K.C. Waugh, Catal. Lett. 30 (1995) 99.
J. Pritchard, T. Catterick and R.K. Gupta, Surf. Sci. 53 (1975) 1.
I.E. Wachs and R.J. Madix, J. Catal. 53 (1978) 208.
M. Balooch, M.J. Cardillo, D.R. Miller and R.E. Stickney, Surf. Sci. 46 (1974) 358.
G. Anger, A. Winkler and K.D. Rendulic, Surf. Sci. 220 (1989) 1.
J.M. Campbell and C.T. Campbell, Surf. Sci. 259 (1991) 1.
B.E. Hayden and C.L.A. Lamont, Chem. Phys. Lett. 160 (1989) 331.
B.E. Hayden and C.L.A. Lamont, Phys. Rev. Lett. 63 (1989) 1823.
B.E. Hayden, D. Lackey and J. Schott, Surf. Sci. 239 (1990) 119.
P.B. Rasmussen, P.M. Holmblad, H. Christoffersen, P.A. Taylor and I. Chorkendorff, Surf. Sci. 287/288 (1993) 79.
F. Besenbacher, P.T. Sprunger, L. Raun, L. Olesen, I. Stensgaard and E. Loegsgaard, Topics Catal. 1 (1994) 325.
K.H. Rieder and W. Stocker, Phys. Rev. Lett. 57 (1986) 2548.
A.R. Gregory, A. Gelb and R. Silbey, Surf. Sci. 74 (1978) 497.
M.R. Hand and S. Holloway, J. Chem. Phys. 91 (1989) 7209.
G.R. Darling and S. Holloway, J. Chem. Phys. 101 (1994) 3268.
G. Mills and H. Jonsson, Phys. Rev. Lett. 72 (1994) 1124.
G. Wiesenekker, G.J. Kroes, E.J. Baerends and R.C. Mowrey, J. Chem. Phys. 102 (1995) 3873.
M. Muhler, L.P. Nielsen, E. Tornqvist, B.S. Clausen and H. Topsøe, Catal. Lett. 14 (1992) 241.
G.C. Chinchen, C.M. Hay, H.D. Vandervell and K.C. Waugh, J. Catal. 103 (1987) 79.
K.C. Waugh, Appl. Catal. 43 (1988) 315.
G.J. Millar, C.H. Rochester, S. Bailey and K.C. Waugh, J. Chem. Soc. Faraday Trans. 89 (1993) 1109.
D.L. Roberts and G.L. Griffin, Appl. Surf. Sci. 19 (1984) 298.
D.L. Roberts and G.L. Griffin, J. Catal. 110 (1988) 117.
R.A. Hadden, B. Sakakini, J. Tabatabaei and K.C. Waugh, Catal. Lett. 44 (1997) 145.
P.A. Redhead, Trans. Faraday Soc. 57 (1961) 641.
K.C. Waugh, Elementary Reaction Steps in Heterogeneous Catalysis, Series C: Math. Phys. Sci. 398 (1993) 407.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tabatabaei, J., Sakakini, B., Watson, M. et al. The detailed kinetics of the desorption of hydrogen from polycrystalline copper catalysts. Catalysis Letters 59, 143–149 (1999). https://doi.org/10.1023/A:1019080823616
Issue Date:
DOI: https://doi.org/10.1023/A:1019080823616