-
 Assessment of Some Unsymmetrical Porphyrins as Promising Molecules for Photodynamic Therapy of Cutaneous Disorders
Assessment of Some Unsymmetrical Porphyrins as Promising Molecules for Photodynamic Therapy of Cutaneous Disorders -
 Antenatal Magnesium Sulfate Benefits Female Preterm Infants but Results in Poor Male Outcomes
Antenatal Magnesium Sulfate Benefits Female Preterm Infants but Results in Poor Male Outcomes -
 Alpha-Emitting Radionuclides: Current Status and Future Perspectives
Alpha-Emitting Radionuclides: Current Status and Future Perspectives -
 A Review of Current and Pipeline Drugs for Treatment of Melanoma
A Review of Current and Pipeline Drugs for Treatment of Melanoma -
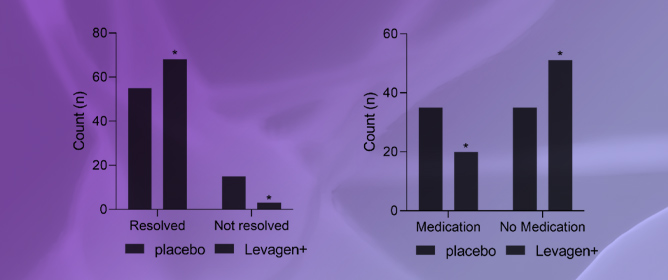 Effectiveness of Palmitoylethanolamide (Levagen+) Compared to a Placebo for Reducing Pain, Duration, and Medication Use during Migraines in Otherwise Healthy Participants—A Double-Blind Randomised Controlled Study
Effectiveness of Palmitoylethanolamide (Levagen+) Compared to a Placebo for Reducing Pain, Duration, and Medication Use during Migraines in Otherwise Healthy Participants—A Double-Blind Randomised Controlled Study
Journal Description
Pharmaceuticals
Pharmaceuticals
is a peer-reviewed, open access journal of medicinal chemistry and related drug sciences, published monthly online by MDPI. The Academy of Pharmaceutical Sciences (APS) is partners of Pharmaceuticals and their members receive a discount on the article processing charge.
- Open Access free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Pharmacology & Pharmacy) / CiteScore - Q2 (Pharmaceutical Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.6 days after submission; acceptance to publication is undertaken in 3.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our editors and authors say about Pharmaceuticals.
- International Electronic Conference on Medicinal Chemistry (https://sciforum.net/series/ecmc/latest)
- Companion journals for Pharmaceuticals include: Pharmacoepidemiology, Psychoactives and Drugs and Drug Candidates.
Impact Factor:
4.6 (2022);
5-Year Impact Factor:
4.9 (2022)
Latest Articles
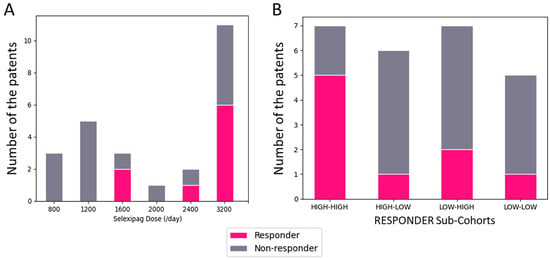
Switching from Beraprost to Selexipag in the Treatment of Pulmonary Arterial Hypertension: Insights from a Phase IV Study of the Japanese Registry (The EXCEL Study: EXChange from bEraprost to seLexipag Study)
Pharmaceuticals 2024, 17(5), 555; https://doi.org/10.3390/ph17050555 (registering DOI) - 26 Apr 2024
Abstract
Pulmonary arterial hypertension (PAH) remains a significant challenge in cardiology, necessitating advancements in treatment strategies. This study explores the safety and efficacy of transitioning patients from beraprost to selexipag, a novel selective prostacyclin receptor agonist, within a Japanese cohort. Employing a multicenter, open-label,
[...] Read more.
Pulmonary arterial hypertension (PAH) remains a significant challenge in cardiology, necessitating advancements in treatment strategies. This study explores the safety and efficacy of transitioning patients from beraprost to selexipag, a novel selective prostacyclin receptor agonist, within a Japanese cohort. Employing a multicenter, open-label, prospective design, 25 PAH patients inadequately managed on beraprost were switched to selexipag. Key inclusion criteria included ongoing beraprost therapy for ≥3 months, a diagnosis of PAH confirmed by mean pulmonary artery pressure (mPAP) ≥ 25 mmHg, and current treatment with endothelin receptor antagonists and/or phosphodiesterase type 5 inhibitors. Outcomes assessed were changes in hemodynamic parameters (mPAP, cardiac index, pulmonary vascular resistance) and the 6 min walk distance (6-MWD) over 3–6 months. The study found no statistically significant changes in these parameters post-switch. However, a subset of patients, defined as responders, demonstrated improvements in all measured hemodynamic parameters, suggesting a potential benefit in carefully selected patients. The transition was generally well-tolerated with no serious adverse events reported. This investigation underscores the importance of personalized treatment strategies in PAH, highlighting that certain patients may benefit from switching to selexipag, particularly those previously on higher doses of beraprost. Further research is needed to elucidate the predictors of positive response to selexipag and optimize treatment regimens for this complex condition.
Full article
(This article belongs to the Special Issue The 20th Anniversary of Pharmaceuticals—Advances in Medicinal Chemistry)
►
Show Figures
Open AccessArticle
Global Health Priority Box: Discovering Flucofuron as a Promising Antikinetoplastid Compound
by
Carlos J. Bethencourt-Estrella, Atteneri López-Arencibia, Jacob Lorenzo-Morales and José E. Piñero
Pharmaceuticals 2024, 17(5), 554; https://doi.org/10.3390/ph17050554 (registering DOI) - 25 Apr 2024
Abstract
Leishmaniasis, produced by Leishmania spp., and Chagas disease, produced by Trypanosoma cruzi, affect millions of people around the world. The treatments for these pathologies are not entirely effective and produce some side effects. For these reasons, it is necessary to develop new
[...] Read more.
Leishmaniasis, produced by Leishmania spp., and Chagas disease, produced by Trypanosoma cruzi, affect millions of people around the world. The treatments for these pathologies are not entirely effective and produce some side effects. For these reasons, it is necessary to develop new therapies that are more active and less toxic for patients. Some initiatives, such as the one carried out by the Medicines for Malaria Venture, allow for the screening of a large number of compounds of different origins to find alternatives to the lack of trypanocide treatments. In this work, 240 compounds were tested from the Global Health Priority Box (80 compounds with confirmed activity against drug-resistant malaria, 80 compounds for screening against neglected and zoonotic diseases and diseases at risk of drug resistance, and 80 compounds with activity against various vector species) against Trypanosoma cruzi and Leishmania amazonensis. Flucofuron, a compound with activity against vectors and with previous activity reported against Staphylococcus spp. and Schistosoma spp., demonstrates activity against L. amazonensis and T. cruzi and produces programmed cell death in the parasites. Flucofuron seems to be a good candidate for continuing study and proving its use as a trypanocidal agent.
Full article
(This article belongs to the Special Issue Antiparasitics 2023)
Open AccessArticle
Effects of CYP3A5 Genotype on Tacrolimus Pharmacokinetics and Graft-versus-Host Disease Incidence in Allogeneic Hematopoietic Stem Cell Transplantation
by
Daniel N. Marco, Mònica Molina, Ana-María Guio, Judit Julian, Virginia Fortuna, Virginia-Lucila Fabregat-Zaragoza, María-Queralt Salas, Inés Monge-Escartín, Gisela Riu-Viladoms, Esther Carcelero, Joan Ramón Roma, Noemí Llobet, Jordi Arcarons, María Suárez-Lledó, Laura Rosiñol, Francesc Fernández-Avilés, Montserrat Rovira, Mercè Brunet and Carmen Martínez
Pharmaceuticals 2024, 17(5), 553; https://doi.org/10.3390/ph17050553 (registering DOI) - 25 Apr 2024
Abstract
Tacrolimus (Tac) is pivotal in preventing acute graft-versus-host disease (GVHD) after allogeneic hematopoietic stem cell transplantation (alloHSCT). It has been reported that genetic factors, including CYP3A5*3 and CYP3A4*22 polymorphisms, have an impact on Tac metabolism, dose requirement, and response to
[...] Read more.
Tacrolimus (Tac) is pivotal in preventing acute graft-versus-host disease (GVHD) after allogeneic hematopoietic stem cell transplantation (alloHSCT). It has been reported that genetic factors, including CYP3A5*3 and CYP3A4*22 polymorphisms, have an impact on Tac metabolism, dose requirement, and response to Tac. There is limited information regarding this topic in alloHSCT. The CYP3A5 genotype and a low Tac trough concentration/dose ratio (Tac C0/D ratio) can be used to identify fast metabolizers and predict the required Tac dose to achieve target concentrations earlier. We examined 62 Caucasian alloHSCT recipients with a fast metabolizer phenotype (C0/dose ratio ≤ 1.5 ng/mL/mg), assessing CYP3A5 genotypes and acute GVHD incidence. Forty-nine patients (79%) were poor metabolizers (2 copies of the variant *3 allele) and 13 (21%) were CYP3A5 expressers (CYP3A5*1/*1 or CYP3A5*1/*3 genotypes). CYP3A5 expressers had lower C0 at 48 h (3.7 vs. 6.2 ng/mL, p = 0.03) and at 7 days (8.6 vs. 11.4 ng/mL, p = 0.04) after Tac initiation, tended to take longer to reach Tac therapeutic range (11.8 vs. 8.9 days, p = 0.16), and had higher incidence of both global (92.3% vs. 38.8%, p < 0.001) and grade II-IV acute GVHD (61.5% vs. 24.5%, p = 0.008). These results support the adoption of preemptive pharmacogenetic testing to better predict individual Tac initial dose, helping to achieve the therapeutic range and reducing the risk of acute GVHD earlier.
Full article
(This article belongs to the Special Issue Therapeutic Drug Monitoring and Adverse Drug Reactions)
►▼
Show Figures
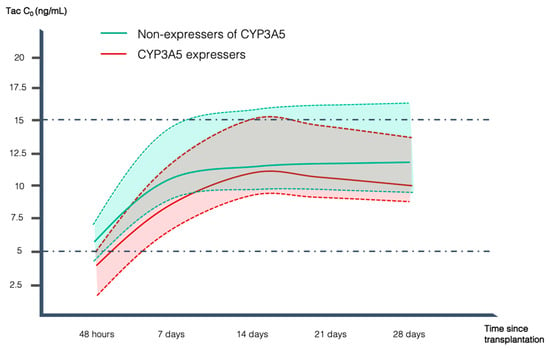
Figure 1
Open AccessArticle
Comparative Investigation of pH–Dependent Availability of Pancreatic Enzyme Preparations In Vitro
by
Amy Todd, Emma Bennett-Huntley, Jonas Rosendahl, Jürgen Schnekenburger and Waldemar Uhl
Pharmaceuticals 2024, 17(5), 552; https://doi.org/10.3390/ph17050552 (registering DOI) - 25 Apr 2024
Abstract
This study aimed to compare different pancreatic enzyme preparations (PEPs) available in Germany regarding particle geometry and size, and to evaluate enzyme activity under physiologically relevant conditions in vitro. Pancreatic endocrine insufficiency is characterized by deficiency of pancreatic enzymes resulting in maldigestion. It
[...] Read more.
This study aimed to compare different pancreatic enzyme preparations (PEPs) available in Germany regarding particle geometry and size, and to evaluate enzyme activity under physiologically relevant conditions in vitro. Pancreatic endocrine insufficiency is characterized by deficiency of pancreatic enzymes resulting in maldigestion. It is orally treated by pancreatic enzyme replacement therapy. The formulations differ in their physical properties and enzyme release behavior, potentially resulting in inconsistent dosages and poor interchangeability of products. A total of 25 products were analyzed for particle size and number of particles per capsule. Enzyme activities of lipase, amylase, and protease were measured by digestion of olive oil emulsion, starch, and casein, respectively. To analyze enzyme release, gastric environments were simulated by incubating PEPs at pH 1, 4, or 5. Duodenal conditions were simulated by subsequent incubation at pH 6. Regarding physical properties and enzyme release kinetics, considerable differences between different PEPs were found. Furthermore, compared to the label claim, excess lipase activity was observed for most products, reaching up to 148%. These in vitro results suggest poor interchangeability of PEPs, potentially explained by physical and release characteristics. Physicians and patients should be aware of the potential gap between label claims and the real-life performance of different PEPs.
Full article
(This article belongs to the Section Pharmaceutical Technology)
►▼
Show Figures
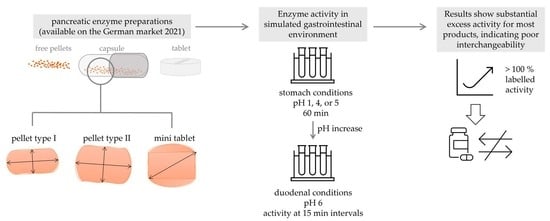
Graphical abstract
Open AccessArticle
In Silico Prediction of New Inhibitors for Kirsten Rat Sarcoma G12D Cancer Drug Target Using Machine Learning-Based Virtual Screening, Molecular Docking, and Molecular Dynamic Simulation Approaches
by
Amar Ajmal, Muhammad Danial, Maryam Zulfat, Muhammad Numan, Sidra Zakir, Chandni Hayat, Khulood Fahad Alabbosh, Magdi E. A. Zaki, Arif Ali and Dongqing Wei
Pharmaceuticals 2024, 17(5), 551; https://doi.org/10.3390/ph17050551 (registering DOI) - 25 Apr 2024
Abstract
Single-point mutations in the Kirsten rat sarcoma (KRAS) viral proto-oncogene are the most common cause of human cancer. In humans, oncogenic KRAS mutations are responsible for about 30% of lung, pancreatic, and colon cancers. One of the predominant mutant KRAS G12D variants is
[...] Read more.
Single-point mutations in the Kirsten rat sarcoma (KRAS) viral proto-oncogene are the most common cause of human cancer. In humans, oncogenic KRAS mutations are responsible for about 30% of lung, pancreatic, and colon cancers. One of the predominant mutant KRAS G12D variants is responsible for pancreatic cancer and is an attractive drug target. At the time of writing, no Food and Drug Administration (FDA) approved drugs are available for the KRAS G12D mutant. So, there is a need to develop an effective drug for KRAS G12D. The process of finding new drugs is expensive and time-consuming. On the other hand, in silico drug designing methodologies are cost-effective and less time-consuming. Herein, we employed machine learning algorithms such as K-nearest neighbor (KNN), support vector machine (SVM), and random forest (RF) for the identification of new inhibitors against the KRAS G12D mutant. A total of 82 hits were predicted as active against the KRAS G12D mutant. The active hits were docked into the active site of the KRAS G12D mutant. Furthermore, to evaluate the stability of the compounds with a good docking score, the top two complexes and the standard complex (MRTX-1133) were subjected to 200 ns MD simulation. The top two hits revealed high stability as compared to the standard compound. The binding energy of the top two hits was good as compared to the standard compound. Our identified hits have the potential to inhibit the KRAS G12D mutation and can help combat cancer. To the best of our knowledge, this is the first study in which machine-learning-based virtual screening, molecular docking, and molecular dynamics simulation were carried out for the identification of new promising inhibitors for the KRAS G12D mutant.
Full article
(This article belongs to the Special Issue Computer-Aided Drug Design and Drug Discovery)
►▼
Show Figures
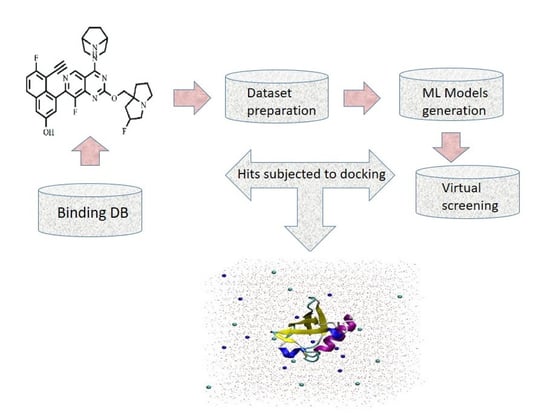
Graphical abstract
Open AccessArticle
Mucoadhesive Budesonide Solution for the Treatment of Pediatric Eosinophilic Esophagitis
by
Antonio Spennacchio, Antonio Lopalco, Giuseppe Francesco Racaniello, Annalisa Cutrignelli, Flavia Maria la Forgia, Sergio Fontana, Fernanda Cristofori, Ruggiero Francavilla, Angela Assunta Lopedota and Nunzio Denora
Pharmaceuticals 2024, 17(5), 550; https://doi.org/10.3390/ph17050550 (registering DOI) - 24 Apr 2024
Abstract
Eosinophilic Esophagitis is an antigen-mediated inflammatory disease characterized by thickening of the esophageal wall, leading to dysphagia, vomiting, reflux, and abdominal pain. This disease can be treated with a therapeutic approach ranging from diet to pharmacological therapy. Jorveza® (budesonide) and Dupixent®
[...] Read more.
Eosinophilic Esophagitis is an antigen-mediated inflammatory disease characterized by thickening of the esophageal wall, leading to dysphagia, vomiting, reflux, and abdominal pain. This disease can be treated with a therapeutic approach ranging from diet to pharmacological therapy. Jorveza® (budesonide) and Dupixent® (dupilumab) are treatments for Eosinophilic Esophagitis approved by the European Medicines Agency in adults but not in children. Budesonide-based extemporaneous oral liquid suspensions could be prepared for pediatric use. The main limit of this formulation is that budesonide needs a longer residence time on the esophageal mucosa to solubilize and diffuse in it to exert its local anti-inflammatory effect. Herein, we propose the development of an extemporaneous mucoadhesive oral budesonide solution for the pediatric population. A liquid vehicle containing hydroxypropyl-beta-cyclodextrin as a complexing agent and carboxymethylcellulose sodium as a mucoadhesive excipient was used to prepare budesonide-based formulations. A stable solution at a concentration of 0.7 mg/mL was successfully prepared and characterized. The formulation showed rheological and mucoadhesive properties suitable for an Eosinophilic Esophagitis local prolonged treatment. In this way, pharmacists can prepare stable budesonide-based mucoadhesive solutions, providing both patients and physicians with a new therapeutic option for Eosinophilic Esophagitis pediatric treatment.
Full article
(This article belongs to the Special Issue Development of Medicines for Rare Pediatric Diseases II)
Open AccessArticle
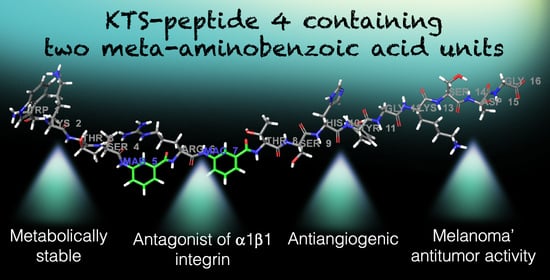
Structure–Activity Relationship of Synthetic Linear KTS-Peptides Containing Meta-Aminobenzoic Acid as Antagonists of α1β1 Integrin with Anti-Angiogenic and Melanoma Anti-Tumor Activities
by
Majdi Saleem Naamneh, Tatjana Momic, Michal Klazas, Julius Grosche, Johannes A. Eble, Cezary Marcinkiewicz, Netaly Khazanov, Hanoch Senderowitz, Amnon Hoffman, Chaim Gilon, Jehoshua Katzhendler and Philip Lazarovici
Pharmaceuticals 2024, 17(5), 549; https://doi.org/10.3390/ph17050549 - 24 Apr 2024
Abstract
To develop peptide drugs targeting integrin receptors, synthetic peptide ligands endowed with well-defined selective binding motifs are necessary. The snake venom KTS-containing disintegrins, which selectively block collagen α1β1 integrin, were used as lead compounds for the synthesis and structure–activity relationship of a series
[...] Read more.
To develop peptide drugs targeting integrin receptors, synthetic peptide ligands endowed with well-defined selective binding motifs are necessary. The snake venom KTS-containing disintegrins, which selectively block collagen α1β1 integrin, were used as lead compounds for the synthesis and structure–activity relationship of a series of linear peptides containing the KTS-pharmacophore and alternating natural amino acids and 3-aminobenzoic acid (MABA). To ensure a better stiffness and metabolic stability, one, two and three MABA residues, were introduced around the KTS pharmacophore motif. Molecular dynamics simulations determined that the solution conformation of MABA peptide 4 is more compact, underwent larger conformational changes until convergence, and spent most of the time in a single cluster. The peptides’ binding affinity has been characterized by an enzyme linked immunosorbent assay in which the most potent peptide 4 inhibited with IC50 of 324 ± 8 µM and 550 ± 45 µM the binding of GST-α1-A domain to collagen IV fragment CB3, and the cell adhesion to collagen IV using α1-overexpressor cells, respectively. Docking studies and MM-GBSA calculations confirmed that peptide 4 binds a smaller region of the integrin near the collagen-binding site and penetrated deeper into the binding site near Trp1. Peptide 4 inhibited tube formation by endothelial cell migration in the Matrigel angiogenesis in vitro assay. Peptide 4 was acutely tolerated by mice, showed stability in human serum, decreased tumor volume and angiogenesis, and significantly increased the survival of mice injected with B16 melanoma cells. These findings propose that MABA-peptide 4 can further serve as an α1β1-integrin antagonist lead compound for further drug optimization in angiogenesis and cancer therapy.
Full article
(This article belongs to the Special Issue Advanced Strategies in Drug Design and Development of Small Molecules as Therapeutic Agents)
►▼
Show Figures
Graphical abstract
Open AccessArticle
NMI-SO2Cl2-Mediated Amide Bond Formation: Facile Synthesis of Some Dihydrotriazolopyrimidine Amide Derivatives as Potential Anti-Inflammatory and Anti-Tubercular Agents
by
Aravinda Babu, Kenchaiah Sunil, Ayyiliath Meleveetil Sajith, Eeda Koti Reddy, Sougata Santra, Grigory V. Zyryanov, Talavara Venkatesh, Somashekara Bhadrachari and Muthipeedika Nibin Joy
Pharmaceuticals 2024, 17(5), 548; https://doi.org/10.3390/ph17050548 - 24 Apr 2024
Abstract
Facile access to some novel biologically relevant dihydrotriazolopyrimidine carboxylic acid-derived amide analogues using NMI/SO2Cl2, and aromatic and aliphatic primary and secondary amines, is reported herein. The role of N-methylimidazole (NMI) as the base and sulfuryl chloride (SO2Cl
[...] Read more.
Facile access to some novel biologically relevant dihydrotriazolopyrimidine carboxylic acid-derived amide analogues using NMI/SO2Cl2, and aromatic and aliphatic primary and secondary amines, is reported herein. The role of N-methylimidazole (NMI) as the base and sulfuryl chloride (SO2Cl2) as the coupling reagent has been effectively realized in accessing these molecules in good to excellent yields. The feasibility of the developed protocol has also been extended to the gram-scale synthesis of N-benzylbenzamide in a 75% yield from benzoic acid and benzyl amine. The newly synthesized compounds were tested via in vitro anti-inflammatory and anti-tubercular activity studies. The compounds 6aa and 6be were found to be the most active anti-inflammatory agents, whereas 6cb and 6ch were found to exhibit promising anti-tubercular potency when compared to other synthesized molecules. The structure–activity relationship (SAR) studies revealed the importance of the presence of electron-donating functionalities in enhancing the anti-inflammatory potential of the newly synthesized molecules. However, the presence of electron-withdrawing substituents was found to be significant for improving their anti-tubercular potency.
Full article
(This article belongs to the Special Issue Design, Synthesis, and Biological Evaluation of Heterocyclic Compounds)
Open AccessArticle
Cholinergic Stimulation Exerts Cardioprotective Effects and Alleviates Renal Inflammatory Responses after Acute Myocardial Infarction in Spontaneous Hypertensive Rats (SHRs)
by
Pamela Nithzi Bricher Choque, Maria Helena Porter, Manuella S. Teixeira, Humberto Dellê, Rosilene Motta Elias, Bruno Durante, Marina Rascio Henriques Dutra, Christine N. Metz, Valentin A. Pavlov and Fernanda M. Consolim Colombo
Pharmaceuticals 2024, 17(5), 547; https://doi.org/10.3390/ph17050547 - 24 Apr 2024
Abstract
Background: In this investigation, we explored the effects of pharmacological cholinergic stimulation on cardiac function and renal inflammation following acute myocardial infarction (AMI) in spontaneously hypertensive rats (SHRs). Methods: Adult male SHRs were randomized into three experimental groups: sham-operated; AMI + Veh (infarcted,
[...] Read more.
Background: In this investigation, we explored the effects of pharmacological cholinergic stimulation on cardiac function and renal inflammation following acute myocardial infarction (AMI) in spontaneously hypertensive rats (SHRs). Methods: Adult male SHRs were randomized into three experimental groups: sham-operated; AMI + Veh (infarcted, treated with vehicle); and AMI + PY (infarcted, treated with the cholinesterase inhibitor, pyridostigmine bromide (PY)—40 mg/kg, once daily for seven days). Rats were euthanized 7 or 30 days post-surgery. The clinical parameters were assessed on the day before euthanasia. Subsequent to euthanasia, blood samples were collected and renal tissues were harvested for histological and gene expression analyses aimed to evaluate inflammation and injury. Results: Seven days post-surgery, the AMI + PY group demonstrated improvements in left ventricular diastolic function and autonomic regulation, and a reduction in renal macrophage infiltration compared to the AMI + Veh group. Furthermore, there was a notable downregulation in pro-inflammatory gene expression and an upregulation in anti-inflammatory gene expression. Analysis 30 days post-surgery showed that PY treatment had a sustained positive effect on renal gene expression, correlated with a decrease in biomarkers, indicative of subclinical kidney injury. Conclusions: Short-term cholinergic stimulation with PY provides both cardiac and renal protection by mitigating the inflammatory response after AMI.
Full article
(This article belongs to the Special Issue Cardiovascular Neuromodulatory Therapy)
►▼
Show Figures
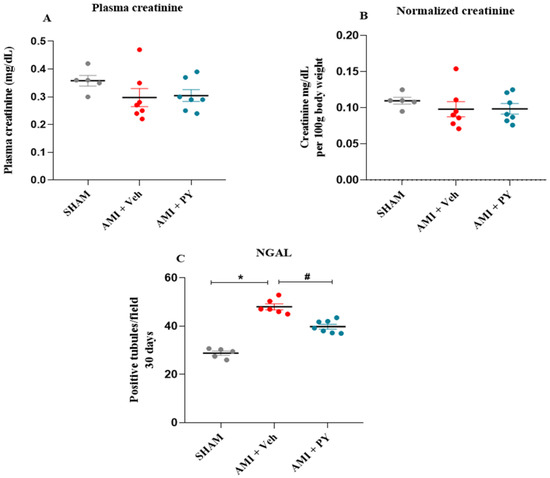
Figure 1
Open AccessArticle
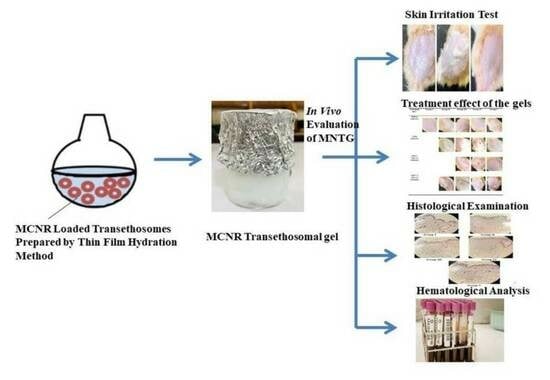
In Vivo Evaluation of Miconazole-Nitrate-Loaded Transethosomal Gel Using a Rat Model Infected with Candida albicans
by
Zara Asghar, Talha Jamshaid, Usama Jamshaid, Asadullah Madni, Naheed Akhtar, Manar O. Lashkar and Heba A. Gad
Pharmaceuticals 2024, 17(5), 546; https://doi.org/10.3390/ph17050546 - 24 Apr 2024
Abstract
Miconazole nitrate (MCNR), an antifungal drug, is used to treat superficial infections. The objective of the current study was to assess the antifungal effectiveness of MCNR-loaded transethosomal gel (MNTG) against Candida albicans in an in vivo rat model. The outcomes were compared with
[...] Read more.
Miconazole nitrate (MCNR), an antifungal drug, is used to treat superficial infections. The objective of the current study was to assess the antifungal effectiveness of MCNR-loaded transethosomal gel (MNTG) against Candida albicans in an in vivo rat model. The outcomes were compared with those of the miconazole nitrate gel (MNG) and marketed Daktarin® cream (2%) based on histopathological and hematological studies. The results of the skin irritation test revealed the safety profile of the MNTG. The MNTG demonstrated the greatest antifungal activity in the histological analysis and the visible restoration of the skin, and the rats revealed an apparent evidence of recovery. Compared to the untreated group, the treated group’s lymphocyte and white blood cells counts increased, but their eosinophil counts decreased. In conclusion, MNTG exhibited the greatest antifungal activity, which might be connected to the improved skin permeability of the transethosome’s nanosized vesicles. Therefore, it could be considered a promising carrier for topical usage and the treatment of cutaneous candidiasis. More clinical research needs to be performed in order to demonstrate its effectiveness and safe usage in humans.
Full article
(This article belongs to the Special Issue Lipid-Based Nanocarriers in Drug Delivery and Theranostic Applications)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Antibacterial, Anti-Biofilm, and Anti-Inflammatory Properties of Gelatin–Chitosan–Moringa-Biopolymer-Based Wound Dressings towards Staphylococcus aureus and Escherichia coli
by
Salma Bessalah, Asim Faraz, Mohamed Dbara, Touhami Khorcheni, Mohamed Hammadi, Daniel Jesuwenu Ajose and Shamsaldeen Ibrahim Saeed
Pharmaceuticals 2024, 17(5), 545; https://doi.org/10.3390/ph17050545 - 23 Apr 2024
Abstract
In contemporary times, the sustained aspiration of bioengineering and biomedical applications is the progressive advancement of materials characterized by biocompatibility and biodegradability. The investigation of the potential applications of polymers as natural and non-hazardous materials has placed significant emphasis on their physicochemical properties.
[...] Read more.
In contemporary times, the sustained aspiration of bioengineering and biomedical applications is the progressive advancement of materials characterized by biocompatibility and biodegradability. The investigation of the potential applications of polymers as natural and non-hazardous materials has placed significant emphasis on their physicochemical properties. Thus, this study was designed to investigate the potential of gelatin–chitosan–moringa leaf extract (G–CH–M) as a novel biomaterial for biomedical applications. The wound-dressing G–CH–M biopolymer was synthesized and characterized. The blood haemolysis, anti-inflammatory, antioxidant, and antibacterial activities of the biopolymer were investigated against Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) bacterial isolates. Our results showed that S. aureus swarming motility was drastically affected. However, the biopolymer had no significant effect on the swarming motility of E. coli. In addition, the biopolymer showed high antibacterial capacities, especially against S. aureus. Plasmid DNA was observed to be effectively protected from oxidative stresses by the biopolymer. Furthermore, the biopolymer exhibited greatly suppressed haemolysis (lower than 2%), notwithstanding the elevated concentration of 50 mg/mL. These results indicated that this novel biopolymer formulation could be further developed for wound care and contamination prevention.
Full article
(This article belongs to the Special Issue Bioactive Substances and Skin Health: The Role of Pharmacy and Nutrition—Properties and Technology)
►▼
Show Figures
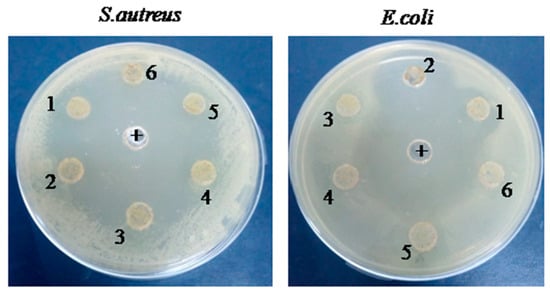
Figure 1
Open AccessArticle
Evaluating the Safety of Herbal Medicine on Renal Function: A Comprehensive Analysis from Six Randomized Controlled Trials Conducted with Four Formulations from Traditional Korean Medicine
by
Jin-Yong Joung and Chang-Gue Son
Pharmaceuticals 2024, 17(5), 544; https://doi.org/10.3390/ph17050544 - 23 Apr 2024
Abstract
The growing popularity of herbal medicine raises concerns about potential nephrotoxicity risks, while limited evidence hinders a comprehensive impact assessment. This study aims to investigate the overall risk features of herbal medicine on kidney injury. We conducted a retrospective analysis on renal function
[...] Read more.
The growing popularity of herbal medicine raises concerns about potential nephrotoxicity risks, while limited evidence hinders a comprehensive impact assessment. This study aims to investigate the overall risk features of herbal medicine on kidney injury. We conducted a retrospective analysis on renal function changes, including blood urea nitrogen (BUN), serum creatinine, and estimated glomerular filtration rate (eGFR), through data from six randomized controlled trials (RCTs) in South Korea. A total of 407 participants (142 males, 265 females) received either one of four different herbal medicines (240 participants) or a placebo (167 participants). When comparing changes in eGFR regarding the mean, 90th-percentile value, and 20% reduction after treatment, there was no significant difference between the herbal-treated and placebo groups. This study provided a helpful reference for examining the safety issues of herbal remedies, especially regarding kidney function.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures
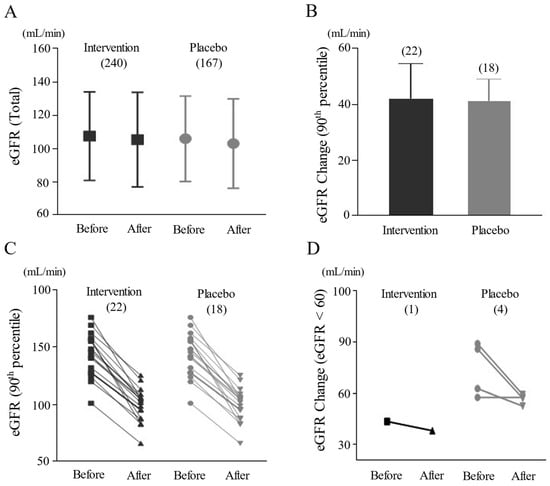
Figure 1
Open AccessArticle
In Silico Design of Potential Small-Molecule Antibiotic Adjuvants against Salmonella typhimurium Ortho Acetyl Sulphydrylase Synthase to Address Antimicrobial Resistance
by
Oluwadunni F. Elebiju, Gbolahan O. Oduselu, Temitope A. Ogunnupebi, Olayinka O. Ajani and Ezekiel Adebiyi
Pharmaceuticals 2024, 17(5), 543; https://doi.org/10.3390/ph17050543 - 23 Apr 2024
Abstract
The inhibition of O-acetyl sulphydrylase synthase isoforms has been reported to represent a promising approach for the development of antibiotic adjuvants. This occurs via the organism developing an unpaired oxidative stress response, causing a reduction in antibiotic resistance in vegetative and swarm
[...] Read more.
The inhibition of O-acetyl sulphydrylase synthase isoforms has been reported to represent a promising approach for the development of antibiotic adjuvants. This occurs via the organism developing an unpaired oxidative stress response, causing a reduction in antibiotic resistance in vegetative and swarm cell populations. This consequently increases the effectiveness of conventional antibiotics at lower doses. This study aimed to predict potential inhibitors of Salmonella typhimurium ortho acetyl sulphydrylase synthase (StOASS), which has lower binding energy than the cocrystalized ligand pyridoxal 5 phosphate (PLP), using a computer-aided drug design approach including pharmacophore modeling, virtual screening, and in silico ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) evaluation. The screening and molecular docking of 4254 compounds obtained from the PubChem database were carried out using AutoDock vina, while a post-screening analysis was carried out using Discovery Studio. The best three hits were compounds with the PubChem IDs 118614633, 135715279, and 155773276, possessing binding affinities of −9.1, −8.9, and −8.8 kcal/mol, respectively. The in silico ADMET prediction showed that the pharmacokinetic properties of the best hits were relatively good. The optimization of the best three hits via scaffold hopping gave rise to 187 compounds, and they were docked against StOASS; this revealed that lead compound 1 had the lowest binding energy (−9.3 kcal/mol) and performed better than its parent compound 155773276. Lead compound 1, with the best binding affinity, has a hydroxyl group in its structure and a change in the core heterocycle of its parent compound to benzimidazole, and pyrimidine introduces a synergistic effect and consequently increases the binding energy. The stability of the best hit and optimized compound at the StOASS active site was determined using RMSD, RMSF, radius of gyration, and SASA plots generated from a molecular dynamics simulation. The MD simulation results were also used to monitor how the introduction of new functional groups of optimized compounds contributes to the stability of ligands at the target active site. The improved binding affinity of these compounds compared to PLP and their toxicity profile, which is predicted to be mild, highlights them as good inhibitors of StOASS, and hence, possible antimicrobial adjuvants.
Full article
(This article belongs to the Special Issue New Perspectives on Chemoinformatics and Drug Design)
►▼
Show Figures
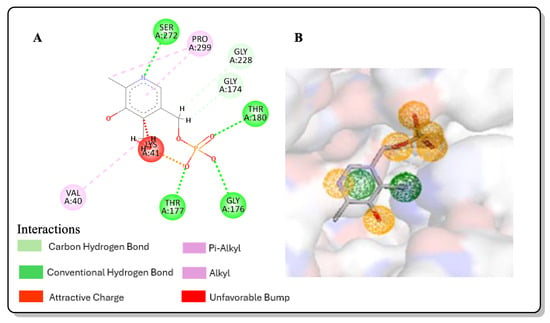
Figure 1
Open AccessSystematic Review
Advancing Postoperative Pain Management in Oral Cancer Patients: A Systematic Review
by
Angelo Michele Inchingolo, Gianna Dipalma, Alessio Danilo Inchingolo, Irene Palumbo, Mariafrancesca Guglielmo, Roberta Morolla, Antonio Mancini and Francesco Inchingolo
Pharmaceuticals 2024, 17(4), 542; https://doi.org/10.3390/ph17040542 - 22 Apr 2024
Abstract
The goal of this review is to shed light on the management of orofacial discomfort after a cancer diagnosis in the head and neck region. A search was conducted on PubMed, Scopus, and Web of Science to identify studies on postoperative pain control
[...] Read more.
The goal of this review is to shed light on the management of orofacial discomfort after a cancer diagnosis in the head and neck region. A search was conducted on PubMed, Scopus, and Web of Science to identify studies on postoperative pain control in oral cancer. The review included open-access research, investigations into pain management, randomized clinical trials, retrospective studies, case-control studies, prospective studies, English-written studies, and full-text publications. Exclusion criteria included animal studies; in vitro studies; off-topic studies; reviews, case reports, letters, or comments; and non-English language. Three reviewers independently accessed databases and assigned a quality rating to the chosen articles. The review explores postoperative pain management in oral cancer patients; highlighting persistent opioid use; the efficacy of adjuvant drugs, such as gabapentin; and a multimodal approach. It emphasizes the need for personalized pain management, recognizing individual pain perception and tailoring interventions. Integrating pharmacological and non-pharmacological strategies is crucial for comprehensive pain management. The review also serves as a guide for future research, emphasizing the need for standardized methodologies and diverse participant populations.
Full article
(This article belongs to the Special Issue Drug Candidates for the Treatment of Oral Cancer and Oral Cancer Pain)
►▼
Show Figures
Figure 1
Open AccessArticle
A Holographic-Type Model in the Description of Polymer–Drug Delivery Processes
by
Irina Nica, Constantin Volovat, Diana Boboc, Ovidiu Popa, Lacramioara Ochiuz, Decebal Vasincu, Vlad Ghizdovat, Maricel Agop, Cristian Constantin Volovat, Corina Lupascu Ursulescu, Cristian Virgil Lungulescu and Simona Ruxandra Volovat
Pharmaceuticals 2024, 17(4), 541; https://doi.org/10.3390/ph17040541 - 22 Apr 2024
Abstract
A unitary model of drug release dynamics is proposed, assuming that the polymer–drug system can be assimilated into a multifractal mathematical object. Then, we made a description of drug release dynamics that implies, via Scale Relativity Theory, the functionality of continuous and undifferentiable
[...] Read more.
A unitary model of drug release dynamics is proposed, assuming that the polymer–drug system can be assimilated into a multifractal mathematical object. Then, we made a description of drug release dynamics that implies, via Scale Relativity Theory, the functionality of continuous and undifferentiable curves (fractal or multifractal curves), possibly leading to holographic-like behaviors. At such a conjuncture, the Schrödinger and Madelung multifractal scenarios become compatible: in the Schrödinger multifractal scenario, various modes of drug release can be “mimicked” (via period doubling, damped oscillations, modulated and “chaotic” regimes), while the Madelung multifractal scenario involves multifractal diffusion laws (Fickian and non-Fickian diffusions). In conclusion, we propose a unitary model for describing release dynamics in polymer–drug systems. In the model proposed, the polymer–drug dynamics can be described by employing the Scale Relativity Theory in the monofractal case or also in the multifractal one.
Full article
(This article belongs to the Section Pharmaceutical Technology)
►▼
Show Figures
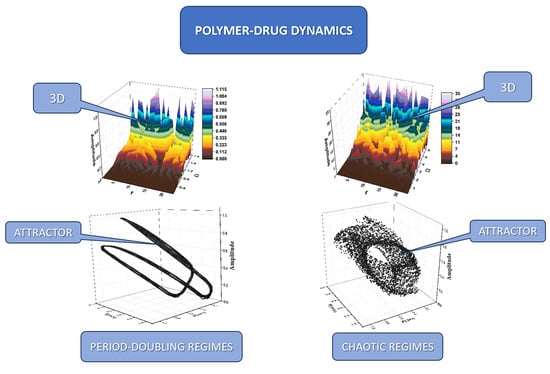
Graphical abstract
Open AccessCorrection
Correction: Nasrullah et al. Omeprazole Prevents Colistin-Induced Nephrotoxicity in Rats: Emphasis on Oxidative Stress, Inflammation, Apoptosis and Colistin Accumulation in Kidneys. Pharmaceuticals 2022, 15, 782
by
Mohammed Z. Nasrullah, Khalid Eljaaly, Thikryat Neamatallah, Usama A. Fahmy, Abdulmohsin J. Alamoudi, Hussain T. Bakhsh and Ashraf B. Abdel-Naim
Pharmaceuticals 2024, 17(4), 540; https://doi.org/10.3390/ph17040540 - 22 Apr 2024
Abstract
Error in Figure [...]
Full article
Open AccessArticle
Nature-Inspired 1-Phenylpyrrolo[2,1-a]isoquinoline Scaffold for Novel Antiproliferative Agents Circumventing P-Glycoprotein-Dependent Multidrug Resistance
by
Alisa A. Nevskaya, Rosa Purgatorio, Tatiana N. Borisova, Alexey V. Varlamov, Lada V. Anikina, Arina Yu. Obydennik, Elena Yu. Nevskaya, Mauro Niso, Nicola A. Colabufo, Antonio Carrieri, Marco Catto, Modesto de Candia, Leonid G. Voskressensky and Cosimo D. Altomare
Pharmaceuticals 2024, 17(4), 539; https://doi.org/10.3390/ph17040539 - 22 Apr 2024
Abstract
Previous studies have shown that some lamellarin-resembling annelated azaheterocyclic carbaldehydes and related imino adducts, sharing the 1-phenyl-5,6-dihydropyrrolo[2,1-a]isoquinoline (1-Ph-DHPIQ) scaffold, are cytotoxic in some tumor cells and may reverse multidrug resistance (MDR) mediated by P-glycoprotein (P-gp). Herein, several novel substituted 1-Ph-DHPIQ derivatives
[...] Read more.
Previous studies have shown that some lamellarin-resembling annelated azaheterocyclic carbaldehydes and related imino adducts, sharing the 1-phenyl-5,6-dihydropyrrolo[2,1-a]isoquinoline (1-Ph-DHPIQ) scaffold, are cytotoxic in some tumor cells and may reverse multidrug resistance (MDR) mediated by P-glycoprotein (P-gp). Herein, several novel substituted 1-Ph-DHPIQ derivatives were synthesized which carry carboxylate groups (COOH, COOEt), nitrile (CN) and Mannich bases (namely, morpholinomethyl derivatives) in the C2 position, as replacements of the already reported aldehyde group. They were evaluated for antiproliferative activity in four tumor cell lines (RD, HCT116, HeLa, A549) and for the ability of selectively inhibiting P-gp-mediated MDR. Lipophilicity descriptors and molecular docking calculations helped us in rationalizing the structure–activity relationships in the P-gp inhibition potency of the investigated 1-Ph-DHPIQs. As a main outcome, a morpholinomethyl Mannich base (8c) was disclosed which proved to be cytotoxic to all the tested tumor cell lines in the low micromolar range (IC50 < 20 μM) and to inhibit in vitro the efflux pumps P-gp and MRP1 responsible for MDR, with IC50s of 0.45 and 12.1 μM, respectively.
Full article
(This article belongs to the Special Issue Medicinal Chemistry of Indole and Quinoline Derivatives: Trends, and Future Directions as Therapeutic Drugs)
►▼
Show Figures
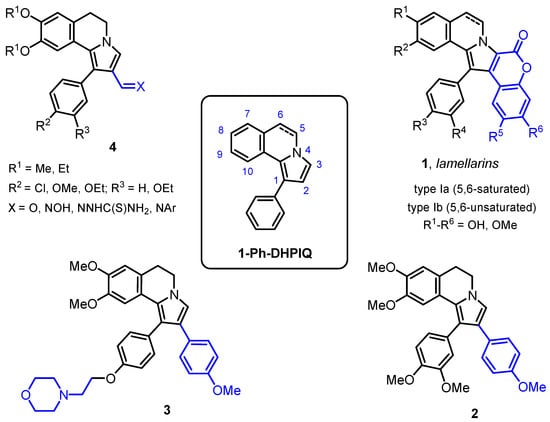
Figure 1
Open AccessArticle
Development of Syringaldehyde as an Agonist of the GLP-1 Receptor to Alleviate Diabetic Disorders in Animal Models
by
Jenpei Lee, Yingxiao Li, Juei-Tang Cheng, I-Min Liu and Kai-Chun Cheng
Pharmaceuticals 2024, 17(4), 538; https://doi.org/10.3390/ph17040538 - 22 Apr 2024
Abstract
The phenolic aldehyde syringaldehyde (SA) has been shown to have an antihyperglycemic effect in diabetic rats due to increased glucose utilization and insulin sensitivity. To understand the direct effect of SA on the GLP-1 receptor, STZ-induced diabetic rats were used. The levels of
[...] Read more.
The phenolic aldehyde syringaldehyde (SA) has been shown to have an antihyperglycemic effect in diabetic rats due to increased glucose utilization and insulin sensitivity. To understand the direct effect of SA on the GLP-1 receptor, STZ-induced diabetic rats were used. The levels of pro-inflammatory cytokines, liver enzymes, and renal function were measured using specific ELISA kits. The mechanisms of SA effects were investigated using CHO-K1 cells, pancreatic Min-6 cells, and cardiomyocyte H9c2 cells. The results indicated that the antihyperglycemic effect of SA in diabetic rats was abolished by blocking the GLP-1 receptor with an antagonist. SA has a direct effect on the GLP-1 receptor when using CHO-K1 cells transfected with the exogenous GLP-1 receptor gene. In addition, SA stimulated insulin production in Min-6 cells by activating GLP-1 receptors. SA caused a dose-dependent rise in GLP-1 receptor mRNA levels in cardiac H9c2 cells. These in vitro results support the notion that SA has a direct effect on the GLP-1 receptor. Otherwise, SA inhibited the increase of pro-inflammatory cytokines, including interleukins and tumor TNF-α, in type 1 diabetic rats in a dose-dependent manner. Moreover, as with liraglutide, SA reduced plasma lipid profiles, including total cholesterol and triglyceride, in mixed diet-induced type 2 diabetic rats. Intriguingly, chronic treatment with SA (as with liraglutide) reversed the functions of both the liver and the kidney in these diabetic rats. SA displayed less efficiency in reducing body weight and food consumption compared to liraglutide. In conclusion, SA effectively activates GLP-1 receptors, resulting in a reduction in diabetic-related complications in rats. Therefore, it is beneficial to develop SA as a chemical agonist for clinical applications in the future.
Full article
(This article belongs to the Section Pharmacology)
►▼
Show Figures
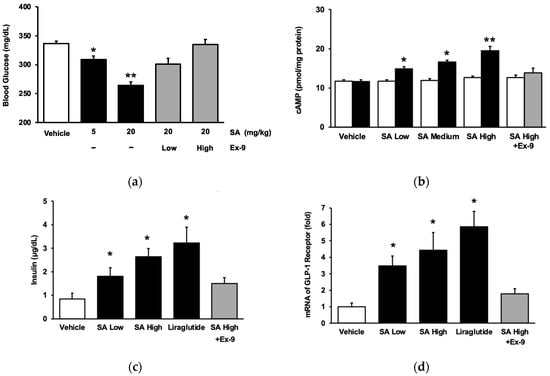
Figure 1
Open AccessReview
Illicit Drugs in Surface Waters: How to Get Fish off the Addictive Hook
by
Halina Falfushynska, Piotr Rychter, Anastasiia Boshtova, Yuliia Faidiuk, Nadiia Kasianchuk and Piotr Rzymski
Pharmaceuticals 2024, 17(4), 537; https://doi.org/10.3390/ph17040537 - 22 Apr 2024
Abstract
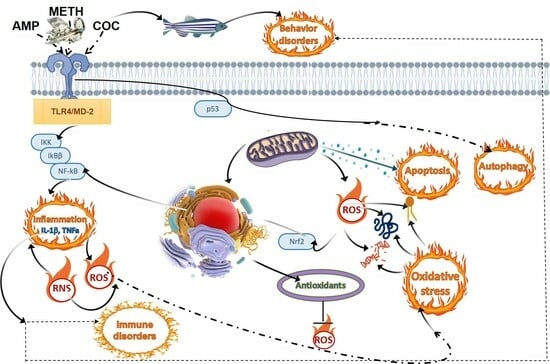
The United Nations World Drug Report published in 2022 alarmed that the global market of illicit drugs is steadily expanding in space and scale. Substances of abuse are usually perceived in the light of threats to human health and public security, while the
[...] Read more.
The United Nations World Drug Report published in 2022 alarmed that the global market of illicit drugs is steadily expanding in space and scale. Substances of abuse are usually perceived in the light of threats to human health and public security, while the environmental aspects of their use and subsequent emissions usually remain less explored. However, as with other human activities, drug production, trade, and consumption of drugs may leave their environmental mark. Therefore, this paper aims to review the occurrence of illicit drugs in surface waters and their bioaccumulation and toxicity in fish. Illicit drugs of different groups, i.e., psychostimulants (methamphetamines/amphetamines, cocaine, and its metabolite benzoylecgonine) and depressants (opioids: morphine, heroin, methadone, fentanyl), can reach the aquatic environment through wastewater discharge as they are often not entirely removed during wastewater treatment processes, resulting in their subsequent circulation in nanomolar concentrations, potentially affecting aquatic biota, including fish. Exposure to such xenobiotics can induce oxidative stress and dysfunction to mitochondrial and lysosomal function, distort locomotion activity by regulating the dopaminergic and glutamatergic systems, increase the predation risk, instigate neurological disorders, disbalance neurotransmission, and produce histopathological alterations in the brain and liver tissues, similar to those described in mammals. Hence, this drugs-related multidimensional harm to fish should be thoroughly investigated in line with environmental protection policies before it is too late. At the same time, selected fish species (e.g., Danio rerio, zebrafish) can be employed as models to study toxic and binge-like effects of psychoactive, illicit compounds.
Full article
(This article belongs to the Special Issue Zebrafish as a Powerful Tool for Drug Discovery 2023)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Synthesis and Pharmacological Characterization of New Photocaged Agonists for Histamine H3 and H4 Receptors
by
Yang Zheng, Meichun Gao, Maikel Wijtmans, Henry F. Vischer and Rob Leurs
Pharmaceuticals 2024, 17(4), 536; https://doi.org/10.3390/ph17040536 - 21 Apr 2024
Abstract
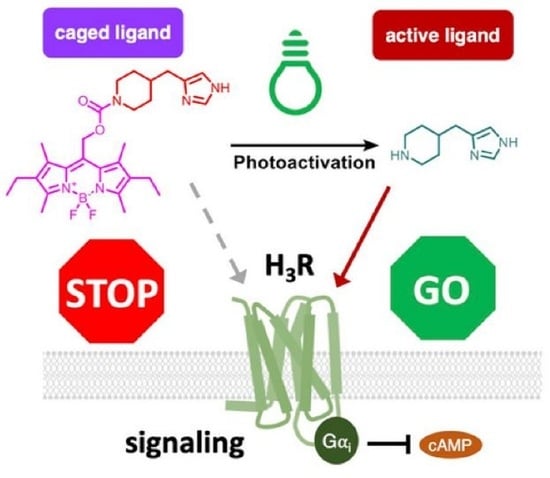
The modulation of biological processes with light-sensitive chemical probes promises precise temporal and spatial control. Yet, the design and synthesis of suitable probes is a challenge for medicinal chemists. This article introduces a photocaging strategy designed to modulate the pharmacology of histamine H
[...] Read more.
The modulation of biological processes with light-sensitive chemical probes promises precise temporal and spatial control. Yet, the design and synthesis of suitable probes is a challenge for medicinal chemists. This article introduces a photocaging strategy designed to modulate the pharmacology of histamine H3 receptors (H3R) and H4 receptors (H4R). Employing the photoremovable group BODIPY as the caging entity for two agonist scaffolds—immepip and 4-methylhistamine—for H3R and H4R, respectively, we synthesized two BODIPY-caged compounds, 5 (VUF25657) and 6 (VUF25678), demonstrating 10–100-fold reduction in affinity for their respective receptors. Notably, the caged H3R agonist, VUF25657, exhibits approximately a 100-fold reduction in functional activity. The photo-uncaging of VUF25657 at 560 nm resulted in the release of immepip, thereby restoring binding affinity and potency in functional assays. This approach presents a promising method to achieve optical control of H3R receptor pharmacology.
Full article
(This article belongs to the Special Issue Histamine Receptor Ligands in Medicinal Chemistry)
►▼
Show Figures
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Pharmaceuticals Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
22 April 2024
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Medicinal Chemistry”
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Medicinal Chemistry”

19 April 2024
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Pharmaceutical Technology”
Pharmaceuticals | Top 5 Selected Papers in 2022–2023 in the Section “Pharmaceutical Technology”

Topics
Topic in
Antioxidants, IJPB, Molecules, Pharmaceuticals, Plants
Plants Volatile Compounds
Topic Editors: Dario Kremer, Igor Jerković, Valerija DunkićDeadline: 30 April 2024
Topic in
BioMed, Biomedicines, Osteology, Pharmaceuticals, Pharmaceutics
Recent Advancement in Biotechnology and Drug Development Using Cutting-Edge Platforms
Topic Editors: Hani Al-Salami, Armin MooranianDeadline: 31 May 2024
Topic in
Antibiotics, JPM, Pharmaceuticals, Pharmaceutics
Pharmacokinetic and Pharmacodynamic Modelling in Drug Discovery and Development
Topic Editors: Inaki F. Troconiz, Victor Mangas Sanjuán, Maria Garcia-Cremades MiraDeadline: 31 August 2024
Topic in
Cancers, Diseases, Nanomaterials, Pharmaceuticals, Materials
Nanomaterials and Diseases
Topic Editors: Diana Díaz-García, Santiago Gómez-RuizDeadline: 13 October 2024

Conferences
Special Issues
Special Issue in
Pharmaceuticals
Advancements and Challenges of Intrapleural and Peritoneal Drug Delivery
Guest Editors: Agata Mikolajczyk-Martinez, Tanja Khosrawipour, Veria KhosrawipourDeadline: 26 April 2024
Special Issue in
Pharmaceuticals
Current Frames on Cardiotoxicology of New and Old Anticancer Drugs
Guest Editor: Vera Marisa CostaDeadline: 8 May 2024
Special Issue in
Pharmaceuticals
Therapeutic Target Developments in Pancreatic Cancer
Guest Editors: Guishan Xiao, Bin LuDeadline: 12 May 2024
Special Issue in
Pharmaceuticals
Recent Advances in TGF-β Inhibitors for the Therapeutic Management of Cancer and Fibrosis
Guest Editor: David DaniepourDeadline: 31 May 2024
Topical Collections
Topical Collection in
Pharmaceuticals
Old Pharmaceuticals with New Applications
Collection Editor: Massimiliano Tognolini
Topical Collection in
Pharmaceuticals
Drug Discovery and Development for Tropical Diseases (TDs)
Collection Editor: Christophe Dardonville
Topical Collection in
Pharmaceuticals
The Story of Successful Drugs and Recent FDA-Approved Molecules
Collection Editors: Maria Emília De Sousa, Jean Jacques Vanden Eynde, Klaus Kopka, Annie Mayence, Joachim Jose, Guangshun WangConference Reports
Pharmaceuticals 2023, 16(3), 432; https://doi.org/10.3390/ph16030432
Pharmaceuticals 2022, 15(4), 388; https://doi.org/10.3390/ph15040388