Journal Description
Marine Drugs
Marine Drugs
is the leading, peer-reviewed, open access journal on the research, development, and production of biologically and therapeutically active compounds from the sea. Marine Drugs is published monthly online by MDPI. Australia New Zealand Marine Biotechnology Society (ANZMBS) is affiliated with Marine Drugs and its members receive a discount on article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, MEDLINE, PMC, Embase, PubAg, MarinLit, AGRIS, and other databases.
- Journal Rank: JCR - Q1 (Pharmacology & Pharmacy) / CiteScore - Q1 (Pharmacology, Toxicology and Pharmaceutics (miscellaneous))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14 days after submission; acceptance to publication is undertaken in 1.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
5.4 (2022);
5-Year Impact Factor:
5.5 (2022)
Latest Articles
Methylation-GC-MS/FID-Based Glycosidic Linkage Analysis of Unfractionated Polysaccharides in Red Seaweeds
Mar. Drugs 2024, 22(5), 192; https://doi.org/10.3390/md22050192 - 24 Apr 2024
Abstract
Glycosidic linkage analysis was conducted on the unfractionated polysaccharides in alcohol-insoluble residues (AIRs) prepared from six red seaweeds (Gracilariopsis sp., Prionitis sp., Mastocarpus papillatus, Callophyllis sp., Mazzaella splendens, and Palmaria palmata) using GC-MS/FID analysis of partially methylated alditol acetates
[...] Read more.
Glycosidic linkage analysis was conducted on the unfractionated polysaccharides in alcohol-insoluble residues (AIRs) prepared from six red seaweeds (Gracilariopsis sp., Prionitis sp., Mastocarpus papillatus, Callophyllis sp., Mazzaella splendens, and Palmaria palmata) using GC-MS/FID analysis of partially methylated alditol acetates (PMAAs). The cell walls of P. palmata primarily contained mixed-linkage xylans and small amounts of sulfated galactans and cellulose. In contrast, the unfractionated polysaccharides of the other five species were rich in galactans displaying diverse 3,6-anhydro-galactose and galactose linkages with varied sulfation patterns. Different levels of cellulose were also observed. This glycosidic linkage method offers advantages for cellulose analysis over traditional monosaccharide analysis that is known for underrepresenting glucose in crystalline cellulose. Relative linkage compositions calculated from GC-MS and GC-FID measurements showed that anhydro sugar linkages generated more responses in the latter detection method. This improved linkage workflow presents a useful tool for studying polysaccharide structural variations across red seaweed species. Furthermore, for the first time, relative linkage compositions from GC-MS and GC-FID measurements, along with normalized FID and total ion current (TIC) chromatograms without peak assignments, were analyzed using principal component analysis (PCA) as a proof-of-concept demonstration of the technique’s potential to differentiate various red seaweed species.
Full article
(This article belongs to the Special Issue Bioactive Polysaccharides from Seaweeds)
►
Show Figures
Open AccessReview
Novel Bioactive Natural Products from Marine-Derived Penicillium Fungi: A Review (2021–2023)
by
Fang Lv and Yanbo Zeng
Mar. Drugs 2024, 22(5), 191; https://doi.org/10.3390/md22050191 - 23 Apr 2024
Abstract
Marine-derived Penicillium fungi are productive sources of structurally unique and diverse bioactive secondary metabolites, representing a hot topic in natural product research. This review describes structural diversity, bioactivities and statistical research of 452 new natural products from marine-derived Penicillium fungi covering 2021 to
[...] Read more.
Marine-derived Penicillium fungi are productive sources of structurally unique and diverse bioactive secondary metabolites, representing a hot topic in natural product research. This review describes structural diversity, bioactivities and statistical research of 452 new natural products from marine-derived Penicillium fungi covering 2021 to 2023. Sediments are the main sources of marine-derived Penicillium fungi for producing nearly 56% new natural products. Polyketides, alkaloids, and terpenoids displayed diverse biological activities and are the major contributors to antibacterial activity, cytotoxicity, anti-inflammatory and enzyme inhibitory capacities. Polyketides had higher proportions of new bioactive compounds in new compounds than other chemical classes. The characteristics of studies in recent years are presented.
Full article
Open AccessArticle
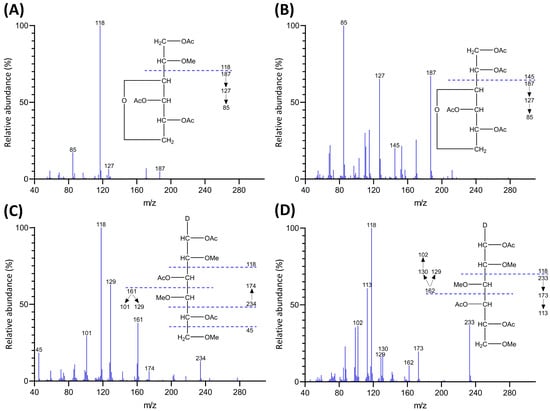
Inhibition Effects and Mechanisms of Marine Compound Mycophenolic Acid Methyl Ester against Influenza A Virus
by
Zihan Wang, Lishan Sun, Hongwei Zhao, Mamadou Dioulde Sow, Yang Zhang and Wei Wang
Mar. Drugs 2024, 22(5), 190; https://doi.org/10.3390/md22050190 - 23 Apr 2024
Abstract
Influenza A virus (IAV) can cause infection and illness in a wide range of animals, including humans, poultry, and swine, and cause annual epidemics, resulting in thousands of deaths and millions of hospitalizations all over the world. Thus, there is an urgent need
[...] Read more.
Influenza A virus (IAV) can cause infection and illness in a wide range of animals, including humans, poultry, and swine, and cause annual epidemics, resulting in thousands of deaths and millions of hospitalizations all over the world. Thus, there is an urgent need to develop novel anti-IAV drugs with high efficiency and low toxicity. In this study, the anti-IAV activity of a marine-derived compound mycophenolic acid methyl ester (MAE) was intensively investigated both in vitro and in vivo. The results showed that MAE inhibited the replication of different influenza A virus strains in vitro with low cytotoxicity. MAE can mainly block some steps of IAV infection post adsorption. MAE may also inhibit viral replication through activating the cellular Akt-mTOR-S6K pathway. Importantly, oral treatment of MAE can significantly ameliorate pneumonia symptoms and reduce pulmonary viral titers, as well as improving the survival rate of mice, and this was superior to the effect of oseltamivir. In summary, the marine compound MAE possesses anti-IAV effects both in vitro and in vivo, which merits further studies for its development into a novel anti-IAV drug in the future.
Full article
(This article belongs to the Special Issue Marine Bioactive Compound Discovery by Combining Virtual with Actual Laboratory Experiments)
►▼
Show Figures
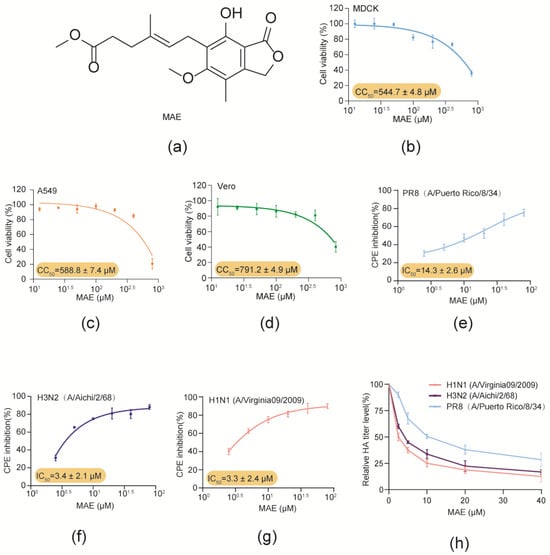
Figure 1
Open AccessArticle
The Discovery of Weddellamycin, a Tricyclic Polyene Macrolactam Antibiotic from an Antarctic Deep-Sea-Derived Streptomyces sp. DSS69, by Heterologous Expression
by
Lu Chen, Kai Liu, Jiali Hong, Zhanzhao Cui, Weijun He, Yemin Wang, Zixin Deng and Meifeng Tao
Mar. Drugs 2024, 22(4), 189; https://doi.org/10.3390/md22040189 - 21 Apr 2024
Abstract
►▼
Show Figures
Polyene macrolactams are a special group of natural products with great diversity, unique structural features, and a wide range of biological activities. Herein, a cryptic gene cluster for the biosynthesis of putative macrolactams was disclosed from a sponge-associated bacterium, Streptomyces sp. DSS69, by
[...] Read more.
Polyene macrolactams are a special group of natural products with great diversity, unique structural features, and a wide range of biological activities. Herein, a cryptic gene cluster for the biosynthesis of putative macrolactams was disclosed from a sponge-associated bacterium, Streptomyces sp. DSS69, by genome mining. Cloning and heterologous expression of the whole biosynthetic gene cluster led to the discovery of weddellamycin, a polyene macrolactam bearing a 23/5/6 ring skeleton. A negative regulator, WdlO, and two positive regulators, WdlA and WdlB, involved in the regulation of weddellamycin production were unraveled. The fermentation titer of weddellamycin was significantly improved by overexpression of wdlA and wdlB and deletion of wdlO. Notably, weddellamycin showed remarkable antibacterial activity against various Gram-positive bacteria including MRSA, with MIC values of 0.10–0.83 μg/mL, and antifungal activity against Candida albicans, with an MIC value of 3.33 μg/mL. Weddellamycin also displayed cytotoxicity against several cancer cell lines, with IC50 values ranging from 2.07 to 11.50 µM.
Full article
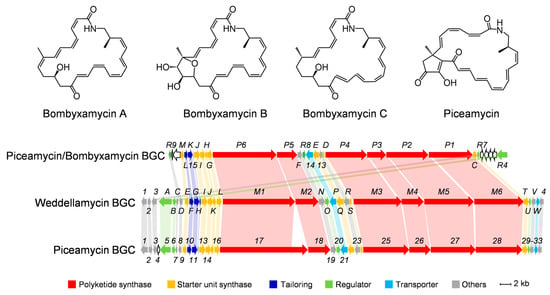
Figure 1
Open AccessArticle
Characterization of Three Polysaccharide-Based Hydrogels Derived from Laminaria japonica and Their Hemostatic Properties
by
Yang Chen, Jinying Shi, Huamai Qiu, Lijun You, Panqi Xu, Rui Rao, Minqian Wu and Ruohan Jia
Mar. Drugs 2024, 22(4), 188; https://doi.org/10.3390/md22040188 - 20 Apr 2024
Abstract
Three Laminaria japonica polysaccharides (LJPs) extracted via water extraction (LJP-W), acid extraction (LJP-A), and enzymatic extraction (LJP-E) were used as raw materials to be cross-linked with chitosan and polyvinyl alcohol to prepare hydrogels. Compared with conventional hydrogel systems, all three types of LJP-based
[...] Read more.
Three Laminaria japonica polysaccharides (LJPs) extracted via water extraction (LJP-W), acid extraction (LJP-A), and enzymatic extraction (LJP-E) were used as raw materials to be cross-linked with chitosan and polyvinyl alcohol to prepare hydrogels. Compared with conventional hydrogel systems, all three types of LJP-based polysaccharide hydrogels exhibited better swelling properties (14 times their original weight) and the absorption ability of simulated body fluid (first 2 h: 6–10%). They also demonstrated better rigidity and mechanical strength. Young’s modulus of LJP-E was 4 times that of the blank. In terms of hemostatic properties, all three polysaccharide hydrogels did not show significant cytotoxic and hemolytic properties. The enzyme- and acid-extracted hydrogels (LJP-Gel-A and LJP-Gel-E) demonstrated better whole-blood coagulant ability compared with the water-extracted hydrogel (LJP-Gel-W), as evidenced by the whole blood coagulation index being half that of LJP-Gel-W. Additionally, the lactate dehydrogenase viabilities of LJP-Gel-A and LJP-Gel-E were significantly higher, at about four and three times those of water extraction, respectively. The above results suggested that LJP-Gel-A and LJP-Gel-E exhibited better blood coagulation capabilities than LJP-Gel-W, due to their enhanced platelet enrichment and adhesion properties. Consequently, these hydrogels are more conducive to promoting coagulation and have good potential for wound hemostasis.
Full article
(This article belongs to the Special Issue Bioactive Polysaccharides from Seaweeds)
►▼
Show Figures
Figure 1
Open AccessArticle
Exploring the Hypocholesterolemic Potential of a Fucus vesiculosus Extract: Omic Insights into Molecular Mechanisms at the Intestinal Level
by
Rebeca André, Rita Pacheco, Hugo M. Santos and Maria Luísa Serralheiro
Mar. Drugs 2024, 22(4), 187; https://doi.org/10.3390/md22040187 - 20 Apr 2024
Abstract
High blood cholesterol levels are a major risk factor for cardiovascular diseases. A purified aqueous extract of Fucus vesiculosus, rich in phlorotannins and peptides, has been described for its potential to inhibit cholesterol biosynthesis and intestinal absorption. In this work, the effect
[...] Read more.
High blood cholesterol levels are a major risk factor for cardiovascular diseases. A purified aqueous extract of Fucus vesiculosus, rich in phlorotannins and peptides, has been described for its potential to inhibit cholesterol biosynthesis and intestinal absorption. In this work, the effect of this extract on intestinal cells’ metabolites and proteins was analysed to gain a deeper understanding of its mode of action on lipids’ metabolism, particularly concerning the absorption and transport of exogenous cholesterol. Caco-2 cells, differentiated into enterocytes, were exposed to the extract, and analysed by untargeted metabolomics and proteomics. The results of the metabolomic analysis showed statistically significant differences in glutathione content of cells exposed to the extract compared to control cells, along with an increased expression of fatty acid amides in exposed cells. A proteomic analysis showed an increased expression in cells exposed to the extract compared to control cells of FAB1 and NPC1, proteins known to be involved in lipid metabolism and transport. To the extent of our knowledge, this study is the first use of untargeted metabolomics and a proteomic analysis to investigate the effects of F. vesiculosus on differentiated Caco-2 cells, offering insights into the molecular mechanism of the extract’s compounds on intestinal cells.
Full article
(This article belongs to the Special Issue Marine-Derived Compounds Applied in Cardiovascular Disease)
►▼
Show Figures
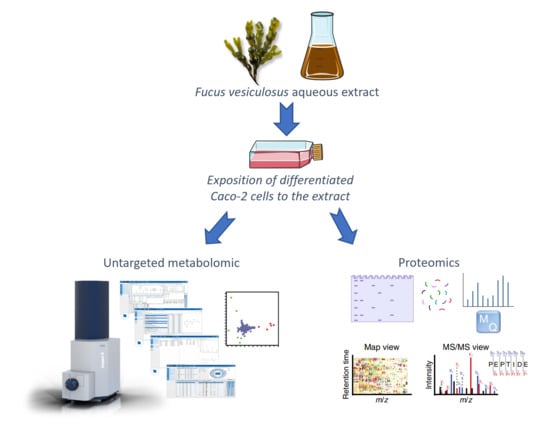
Graphical abstract
Open AccessCommunication
Cytotoxic and Antibacterial Meroterpenoids Isolated from the Marine-Derived Fungus Talaromyces sp. M27416
by
Lingzhi Tang, Jinmei Xia, Zhongwei Chen, Fengjiao Lin, Zongze Shao, Weiyi Wang and Xuan Hong
Mar. Drugs 2024, 22(4), 186; https://doi.org/10.3390/md22040186 - 20 Apr 2024
Abstract
Three novel meroterpenoids, taladrimanins B–D (1–3), were isolated from the marine-derived fungus Talaromyces sp. M27416, alongside three biogenetically related compounds (4–6). We delineated taladrimanin B’s (1) structure using HRESIMS and NMR, confirmed its
[...] Read more.
Three novel meroterpenoids, taladrimanins B–D (1–3), were isolated from the marine-derived fungus Talaromyces sp. M27416, alongside three biogenetically related compounds (4–6). We delineated taladrimanin B’s (1) structure using HRESIMS and NMR, confirmed its configuration via quantum chemical NMR analysis and DP4+ methodology, and verified it through X-ray crystallography. ECD calculations determined the absolute configuration of compound 1, while comparative NMR and ECD analyses elucidated the absolute configurations of 2 and 3. These compounds are drimane-type meroterpenoids with a C10 polyketide unit (8R-configuration). We proposed a biosynthetic pathway and noted that compound 1 showed cytotoxic activity against MKN-45 and 5637 cell lines and selective antibacterial effects against Staphylococcus aureus CICC 10384.
Full article
(This article belongs to the Section Structural Studies on Marine Natural Products)
►▼
Show Figures
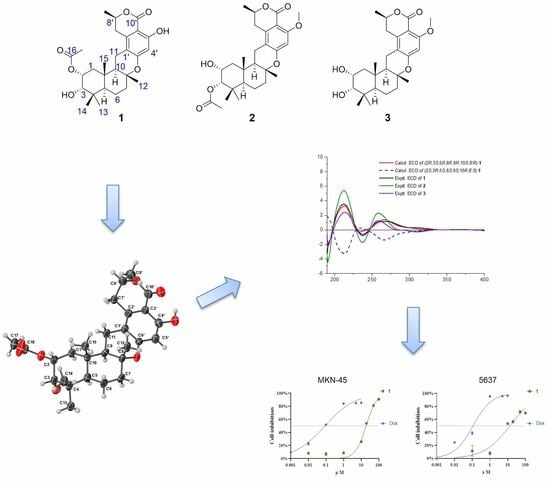
Graphical abstract
Open AccessArticle
Zeaxanthin epoxidase 3 Knockout Mutants of the Model Diatom Phaeodactylum tricornutum Enable Commercial Production of the Bioactive Carotenoid Diatoxanthin
by
Cecilie Græsholt, Tore Brembu, Charlotte Volpe, Zdenka Bartosova, Manuel Serif, Per Winge and Marianne Nymark
Mar. Drugs 2024, 22(4), 185; https://doi.org/10.3390/md22040185 - 19 Apr 2024
Abstract
Carotenoids are pigments that have a range of functions in human health. The carotenoid diatoxanthin is suggested to have antioxidant, anti-inflammatory and chemo-preventive properties. Diatoxanthin is only produced by a few groups of microalgae, where it functions in photoprotection. Its large-scale production in
[...] Read more.
Carotenoids are pigments that have a range of functions in human health. The carotenoid diatoxanthin is suggested to have antioxidant, anti-inflammatory and chemo-preventive properties. Diatoxanthin is only produced by a few groups of microalgae, where it functions in photoprotection. Its large-scale production in microalgae is currently not feasible. In fact, rapid conversion into the inactive pigment diadinoxanthin is triggered when cells are removed from a high-intensity light source, which is the case during large-scale harvesting of microalgae biomass. Zeaxanthin epoxidase (ZEP) 2 and/or ZEP3 have been suggested to be responsible for the back-conversion of high-light-accumulated diatoxanthin to diadinoxanthin in low-light diatoms. Using CRISPR/Cas9 gene editing technology, we knocked out the ZEP2 and ZEP3 genes in the marine diatom Phaeodactylum tricornutum to investigate their role in the diadinoxanthin–diatoxanthin cycle and determine if one of the mutant strains could function as a diatoxanthin production line. Light-shift experiments proved that ZEP3 encodes the enzyme converting diatoxanthin to diadinoxanthin in low light. Loss of ZEP3 caused the high-light-accumulated diatoxanthin to be stable for several hours after the cultures had been returned to low light, suggesting that zep3 mutant strains could be suitable as commercial production lines of diatoxanthin.
Full article
(This article belongs to the Special Issue Marine Anti-inflammatory and Antioxidant Agents 3.0)
►▼
Show Figures
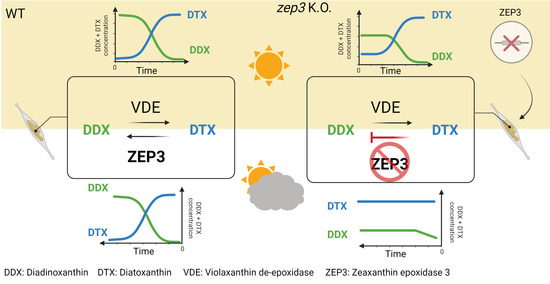
Graphical abstract
Open AccessArticle
Chemical Synthesis of Fucosylated Chondroitin Sulfate Tetrasaccharide with Fucosyl Branch at the 6-OH of GalNAc Residue
by
Changlun Lv, Xiaona Li, Guoqing Yang, Haijiao Chen and Chunxia Li
Mar. Drugs 2024, 22(4), 184; https://doi.org/10.3390/md22040184 - 19 Apr 2024
Abstract
Fucosylated chondroitin sulfate is a unique glycosaminoglycan isolated from sea cucumbers, with excellent anticoagulant activity. The fucosyl branch in FCS is generally located at the 3-OH of D-glucuronic acid but, recently, a novel structure with α-L-fucose linked to the 6-OH
[...] Read more.
Fucosylated chondroitin sulfate is a unique glycosaminoglycan isolated from sea cucumbers, with excellent anticoagulant activity. The fucosyl branch in FCS is generally located at the 3-OH of D-glucuronic acid but, recently, a novel structure with α-L-fucose linked to the 6-OH of N-acetyl-galactosamine has been found. Here, using functionalized monosaccharide building blocks, we prepared novel FCS tetrasaccharides with fucosyl branches both at the 6-OH of GalNAc and 3-OH of GlcA. In the synthesis, the protective group strategy of selective O-sulfation, as well as stereoselective glycosylation, was established, which enabled the efficient synthesis of the specific tetrasaccharide compounds. This research enriches knowledge on the structural types of FCS oligosaccharides and facilitates the exploration of the structure–activity relationship in the future.
Full article
(This article belongs to the Section Synthesis and Medicinal Chemistry of Marine Natural Products)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Meroterpenoids from Marine Sponge Hyrtios sp. and Their Anticancer Activity against Human Colorectal Cancer Cells
by
Jie Wang, Yue-Lu Yan, Xin-Yi Yu, Jia-Yan Pan, Xin-Lian Liu, Li-Li Hong and Bin Wang
Mar. Drugs 2024, 22(4), 183; https://doi.org/10.3390/md22040183 - 19 Apr 2024
Abstract
Two new meroterpenoids, hyrtamide A (1) and hyrfarnediol A (2), along with two known ones, 3-farnesyl-4-hydroxybenzoic acid methyl ester (3) and dictyoceratin C (4), were isolated from a South China Sea sponge Hyrtios sp. Their
[...] Read more.
Two new meroterpenoids, hyrtamide A (1) and hyrfarnediol A (2), along with two known ones, 3-farnesyl-4-hydroxybenzoic acid methyl ester (3) and dictyoceratin C (4), were isolated from a South China Sea sponge Hyrtios sp. Their structures were elucidated by NMR and MS data. Compounds 2–4 exhibited weak cytotoxicity against human colorectal cancer cells (HCT-116), showing IC50 values of 41.6, 45.0, and 37.3 μM, respectively. Furthermore, compounds 3 and 4 significantly suppressed the invasion of HCT-116 cells while also downregulating the expression of vascular endothelial growth factor receptor 1 (VEGFR-1) and vimentin proteins, which are key markers associated with angiogenesis and epithelial–mesenchymal transition (EMT). Our findings suggest that compounds 3 and 4 may exert their anti-invasive effects on tumor cells by inhibiting the expression of VEGFR-1 and impeding the process of EMT.
Full article
(This article belongs to the Special Issue Discovery of Marine Natural Products in China: Selected Papers from the 16th National Annual Conference and 2023 International Symposium on Marine Drugs (16-NASMD) Conference)
►▼
Show Figures
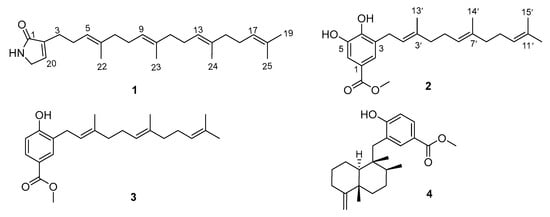
Figure 1
Open AccessReview
Statistical Tools to Optimize the Recovery of Bioactive Compounds from Marine Byproducts
by
Zenebe Tadesse Tsegay, Sofia Agriopoulou, Moufida Chaari, Slim Smaoui and Theodoros Varzakas
Mar. Drugs 2024, 22(4), 182; https://doi.org/10.3390/md22040182 - 18 Apr 2024
Abstract
Techniques for extracting important bioactive molecules from seafood byproducts, viz., bones, heads, skin, frames, fins, shells, guts, and viscera, are receiving emphasis due to the need for better valorization. Employing green extraction technologies for efficient and quality production of these bioactive molecules is
[...] Read more.
Techniques for extracting important bioactive molecules from seafood byproducts, viz., bones, heads, skin, frames, fins, shells, guts, and viscera, are receiving emphasis due to the need for better valorization. Employing green extraction technologies for efficient and quality production of these bioactive molecules is also strictly required. Hence, understanding the extraction process parameters to effectively design an applicable optimization strategy could enable these improvements. In this review, statistical optimization strategies applied for the extraction process parameters of obtaining bioactive molecules from seafood byproducts are focused upon. The type of experimental designs and techniques applied to criticize and validate the effects of independent variables on the extraction output are addressed. Dominant parameters studied were the enzyme/substrate ratio, pH, time, temperature, and power of extraction instruments. The yield of bioactive compounds, including long-chain polyunsaturated fatty acids, amino acids, peptides, enzymes, gelatine, collagen, chitin, vitamins, polyphenolic constituents, carotenoids, etc., were the most studied responses. Efficiency and/or economic and quality considerations and their selected optimization strategies that favor the production of potential bioactive molecules were also reviewed.
Full article
(This article belongs to the Special Issue Sustainable Valorization of Seafood By-Products through Recovery of Valuable Bioactive Compounds)
►▼
Show Figures
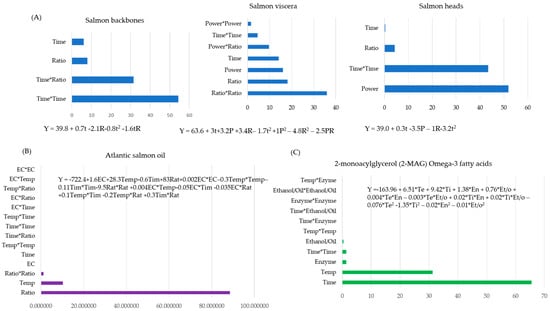
Figure 1
Open AccessArticle
Discovery of a Novel Chromone Enantiomer and the Precursors of Nonactic Acid from the Coral-Reef-Derived Streptomyces sp. SCSIO 66814
by
Wenping Ding, Yanqun Li, Xingyu Li, Jiajia Yin, Songbiao Shi, Xinpeng Tian, Si Zhang and Hao Yin
Mar. Drugs 2024, 22(4), 181; https://doi.org/10.3390/md22040181 - 17 Apr 2024
Abstract
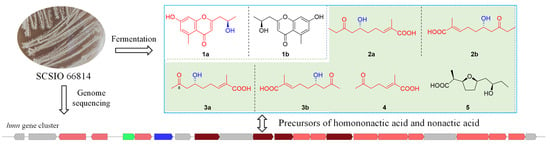
Three pairs of enantiomers (1–3)—the new 12R-aloesol (1a) and two new fatty acids (2 and 3)—and one new natural product (4) together three known compounds (5–7) were
[...] Read more.
Three pairs of enantiomers (1–3)—the new 12R-aloesol (1a) and two new fatty acids (2 and 3)—and one new natural product (4) together three known compounds (5–7) were isolated from a coral-reef-derived Streptomyces sp. SCSIO 66814. Their structures were determined through extensive spectroscopic analysis, chiral analysis, and single-crystal X-ray diffraction data. Compounds 2 and 3 were presumed to be intermediates for further generating homononactic acid (5) and nonactic acid, and the latter two molecules were able to act as precursors to form macrotetrolides with remarkable biological activity. The isolation of related precursors, compounds 2–5, provided more evidence to support the proposal of a plausible biosynthetic pathway for nonactic acid and its homologs. Additionally, (+)-1 exhibited a weak activity against DPPH radicals.
Full article
(This article belongs to the Special Issue Genome Mining and Drug Discovery of Marine and Halophilic Microorganisms)
►▼
Show Figures
Graphical abstract
Open AccessReview
Marine-Derived Metabolites Act as Promising Antifungal Agents
by
Sijin Hang, Hui Lu and Yuanying Jiang
Mar. Drugs 2024, 22(4), 180; https://doi.org/10.3390/md22040180 - 17 Apr 2024
Abstract
The incidence of invasive fungal diseases (IFDs) is on the rise globally, particularly among immunocompromised patients, leading to significant morbidity and mortality. Current clinical antifungal agents, such as polyenes, azoles, and echinocandins, face increasing resistance from pathogenic fungi. Therefore, there is a pressing
[...] Read more.
The incidence of invasive fungal diseases (IFDs) is on the rise globally, particularly among immunocompromised patients, leading to significant morbidity and mortality. Current clinical antifungal agents, such as polyenes, azoles, and echinocandins, face increasing resistance from pathogenic fungi. Therefore, there is a pressing need for the development of novel antifungal drugs. Marine-derived secondary metabolites represent valuable resources that are characterized by varied chemical structures and pharmacological activities. While numerous compounds exhibiting promising antifungal activity have been identified, a comprehensive review elucidating their specific underlying mechanisms remains lacking. In this review, we have compiled a summary of antifungal compounds derived from marine organisms, highlighting their diverse mechanisms of action targeting various fungal cellular components, including the cell wall, cell membrane, mitochondria, chromosomes, drug efflux pumps, and several biological processes, including vesicular trafficking and the growth of hyphae and biofilms. This review is helpful for the subsequent development of antifungal drugs due to its summary of the antifungal mechanisms of secondary metabolites from marine organisms.
Full article
(This article belongs to the Topic Antimicrobial Agents and Nanomaterials)
►▼
Show Figures
Figure 1
Open AccessArticle
Effect of Post-Extraction Ultrasonication on Compositional Features and Antioxidant Activities of Enzymatic/Alkaline Extracts of Palmaria palmata
by
Sakhi Ghelichi, Ann-Dorit Moltke Sørensen, Mona Hajfathalian and Charlotte Jacobsen
Mar. Drugs 2024, 22(4), 179; https://doi.org/10.3390/md22040179 - 17 Apr 2024
Abstract
Palmaria palmata is a viable source of nutrients with bioactive properties. The present study determined the potential role of post-extraction ultrasonication on some compositional features and antioxidant properties of enzymatic/alkaline extracts of P. palmata (EAEP). No significant difference was detected in terms of
[...] Read more.
Palmaria palmata is a viable source of nutrients with bioactive properties. The present study determined the potential role of post-extraction ultrasonication on some compositional features and antioxidant properties of enzymatic/alkaline extracts of P. palmata (EAEP). No significant difference was detected in terms of protein content and recovery, as well as the amino acid composition of the extracts. The nitrogen-to-protein conversion factor of 5 was found to be too high for the seaweed and EAEP. The extracts sonicated by bath for 10 min and not sonicated showed the highest and lowest total phenolic contents (p < 0.05), respectively. The highest radical scavenging and lowest metal-chelating activities were observed for the non-sonicated sample, as evidenced by IC50 values. The extract sonicated by bath for 10 min showed the most favorable in vitro antioxidant properties since its radical scavenging was not significantly different from that of the not-sonicated sample (p > 0.05). In contrast, its metal-chelating activity was significantly higher (p < 0.05). To conclude, post-extraction ultrasonication by an ultrasonic bath for 10 min is recommended to increase phenolic content and improve the antioxidant properties of EAEP.
Full article
(This article belongs to the Section Marine-Derived Ingredients for Drugs, Cosmeceuticals and Nutraceuticals)
►▼
Show Figures
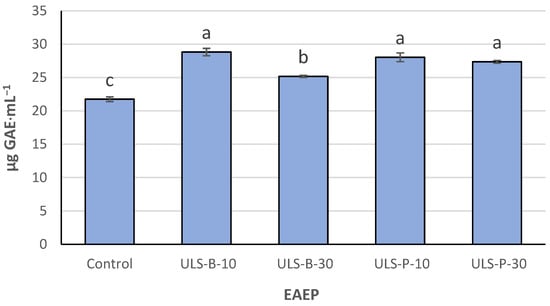
Figure 1
Open AccessArticle
Pharmacokinetics and Safety of Lurbinectedin Administrated with Itraconazole in Cancer Patients: A Drug–Drug Interaction Study
by
Irene Moreno, Tatiana Hernández, Emiliano Calvo, Salvador Fudio, Carmen Kahatt, Sara Martínez, Jorge Luis Iglesias, Román Octavio Calafati, Laura Pérez-Ramos, Lola Montilla, Ali Zeaiter and Rubin Lubomirov
Mar. Drugs 2024, 22(4), 178; https://doi.org/10.3390/md22040178 - 16 Apr 2024
Abstract
This open-label, two-part, phase Ib drug–drug interaction study investigated whether the pharmacokinetic (PK) and safety profiles of lurbinectedin (LRB), a marine-derived drug, are affected by co-administration of itraconazole (ITZ), a strong CYP3A4 inhibitor, in adult patients with advanced solid tumors. In Part A,
[...] Read more.
This open-label, two-part, phase Ib drug–drug interaction study investigated whether the pharmacokinetic (PK) and safety profiles of lurbinectedin (LRB), a marine-derived drug, are affected by co-administration of itraconazole (ITZ), a strong CYP3A4 inhibitor, in adult patients with advanced solid tumors. In Part A, three patients were sequentially assigned to Sequence 1 (LRB 0.8 mg/m2, 1-h intravenous [IV] + ITZ 200 mg/day oral in Cycle 1 [C1] and LRB alone 3.2 mg/m2, 1 h, IV in Cycle 2 [C2]). In Part B, 11 patients were randomized (1:1) to receive either Sequence 1 (LRB at 0.9 mg/m2 + ITZ in C1 and LRB alone in C2) or Sequence 2 (LRB alone in C1 and LRB + ITZ in C2). Eleven patients were evaluable for PK analysis: three in Part A and eight in Part B (four per sequence). The systemic total exposure of LRB increased with ITZ co-administration: 15% for Cmax, area under the curve (AUC) 2.4-fold for AUC0–t and 2.7-fold for AUC0–∞. Co-administration with ITZ produced statistically significant modifications in the unbound plasma LRB PK parameters. The LRB safety profile was consistent with the toxicities described in previous studies. Co-administration with multiple doses of ITZ significantly altered LRB systemic exposure. Hence, to avoid LRB overexposure when co-administered with strong CYP3A4 inhibitors, an LRB dose reduction proportional to CL reduction should be applied.
Full article
(This article belongs to the Section Marine Pharmacology)
►▼
Show Figures
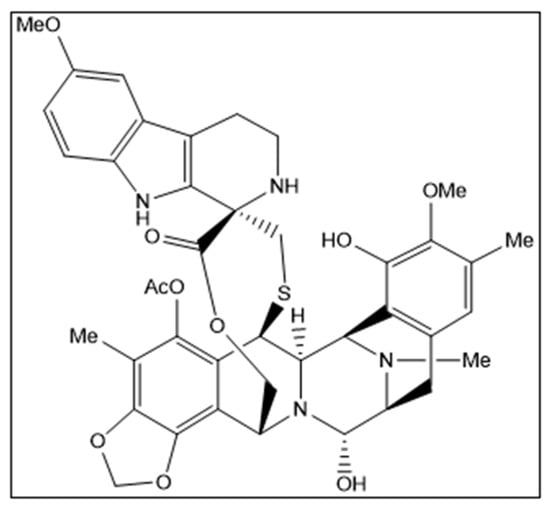
Figure 1
Open AccessArticle
2,3-Dimethoxycinnamic Acid from a Marine Actinomycete, a Promising Quorum Sensing Inhibitor in Chromobacterium violaceum
by
Yanqun Li, Wenping Ding, Jiajia Yin, Xingyu Li, Xinpeng Tian, Zhihui Xiao, Fazuo Wang and Hao Yin
Mar. Drugs 2024, 22(4), 177; https://doi.org/10.3390/md22040177 - 16 Apr 2024
Abstract
An ethyl acetate extract of a marine actinomycete strain, Nocardiopsis mentallicus SCSIO 53858, isolated from a deep-sea sediment sample in the South China Sea, exhibited anti-quorum-sensing (QS) activity against Chromobacterium violaceum CV026. Guided by the anti-QS activity, a novel active compound was isolated
[...] Read more.
An ethyl acetate extract of a marine actinomycete strain, Nocardiopsis mentallicus SCSIO 53858, isolated from a deep-sea sediment sample in the South China Sea, exhibited anti-quorum-sensing (QS) activity against Chromobacterium violaceum CV026. Guided by the anti-QS activity, a novel active compound was isolated and purified from the extract and was identified as 2,3-dimethoxycinnamic acid (2,3-DCA) through spectral data analysis. At a concentration of 150 μg/mL, 2,3-DCA exhibited robust inhibitory effects on three QS-regulated traits of C. violaceum CV026: violacein production, swarming motility, and biofilm formation, with inhibition rates of 73.9%, 65.9%, and 37.8%, respectively. The quantitative reverse transcription polymerase chain reaction results indicated that 2,3-DCA can disrupt the QS system in C. violaceum CV026 by effectively suppressing the expression of QS-related genes, including cviR, vioA, vioB, and vioE. Molecular docking analysis revealed that 2,3-DCA hinders the QS system by competitively binding to the same binding pocket on the CviR receptor as the natural signal molecule N-hexanoyl-L-homoserine lactone. Collectively, these findings suggest that 2,3-DCA exhibits promising potential as an inhibitor of QS systems, providing a potential solution to the emerging problem of bacterial resistance.
Full article
(This article belongs to the Section Marine Biotechnology Related to Drug Discovery or Production)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Automated Patch Clamp for the Detection of Tetrodotoxin in Pufferfish Samples
by
Mònica Campàs, Jaume Reverté, Àngels Tudó, Mounira Alkassar, Jorge Diogène and Francesc X. Sureda
Mar. Drugs 2024, 22(4), 176; https://doi.org/10.3390/md22040176 - 15 Apr 2024
Abstract
Tetrodotoxin (TTX) is a marine toxin responsible for many intoxications around the world. Its presence in some pufferfish species and, as recently reported, in shellfish, poses a serious health concern. Although TTX is not routinely monitored, there is a need for fast, sensitive,
[...] Read more.
Tetrodotoxin (TTX) is a marine toxin responsible for many intoxications around the world. Its presence in some pufferfish species and, as recently reported, in shellfish, poses a serious health concern. Although TTX is not routinely monitored, there is a need for fast, sensitive, reliable, and simple methods for its detection and quantification. In this work, we describe the use of an automated patch clamp (APC) system with Neuro-2a cells for the determination of TTX contents in pufferfish samples. The cells showed an IC50 of 6.4 nM for TTX and were not affected by the presence of muscle, skin, liver, and gonad tissues of a Sphoeroides pachygaster specimen (TTX-free) when analysed at 10 mg/mL. The LOD achieved with this technique was 0.05 mg TTX equiv./kg, which is far below the Japanese regulatory limit of 2 mg TTX equiv./kg. The APC system was applied to the analysis of extracts of a Lagocephalus sceleratus specimen, showing TTX contents that followed the trend of gonads > liver > skin > muscle. The APC system, providing an in vitro toxicological approach, offers the advantages of being sensitive, rapid, and reliable for the detection of TTX-like compounds in seafood.
Full article
(This article belongs to the Special Issue Marine Biotoxins 3.0)
►▼
Show Figures
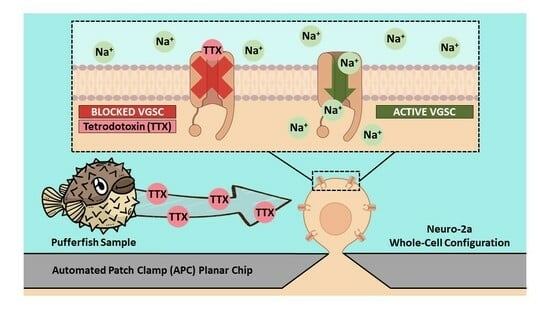
Graphical abstract
Open AccessArticle
Ethanol Extract of Limonium bicolor Improves Dextran Sulfate Sodium-Induced Ulcerative Colitis by Alleviating Inflammation and Restoring Gut Microbiota Dysbiosis in Mice
by
Wei Jia, Siyu Yu, Xi Liu, Qingqing Le, Xiwen He, Lutao Yu, Jianlin He, Longhe Yang and Huiyuan Gao
Mar. Drugs 2024, 22(4), 175; https://doi.org/10.3390/md22040175 - 15 Apr 2024
Abstract
Ulcerative colitis (UC) is a kind of inflammatory bowel condition characterized by inflammation within the mucous membrane, rectal bleeding, diarrhea, and pain experienced in the abdominal region. Existing medications for UC have limited treatment efficacy and primarily focus on symptom relief. Limonium bicolor
[...] Read more.
Ulcerative colitis (UC) is a kind of inflammatory bowel condition characterized by inflammation within the mucous membrane, rectal bleeding, diarrhea, and pain experienced in the abdominal region. Existing medications for UC have limited treatment efficacy and primarily focus on symptom relief. Limonium bicolor (LB), an aquatic traditional Chinese medicine (TCM), exerts multi-targeted therapeutic effects with few side effects and is used to treat anemia and hemostasis. Nevertheless, the impact of LB on UC and its mechanism of action remain unclear. Therefore, the objective of this study was to investigate the anti-inflammatory effects and mechanism of action of ethanol extract of LB (LBE) in lipopolysaccharide-induced RAW 264.7 macrophages and dextran sulfate sodium (DSS)-induced UC. The results showed that LBE suppressed the secretion of cytokines in LPS-stimulated RAW 264.7 cells in a dose-dependent manner. LBE had protective effects against DSS-induced colitis in mice, decreased the disease activity index (DAI) score, alleviated symptoms, increased colon length, and improved histological characteristics, thus having protective effects against DSS-induced colitis in mice. In addition, it reversed disturbances in the abundance of proteobacteria and probiotics such as Lactobacillus and Blautia in mice with DSS-induced UC. Based on the results of network pharmacology analysis, we identified four main compounds in LBE that are associated with five inflammatory genes (Ptgs2, Plg, Ppar-γ, F2, and Gpr35). These results improve comprehension of the biological activity and functionality of LB and may facilitate the development of LB-based compounds for the treatment of UC.
Full article
(This article belongs to the Special Issue Marine-Sourced Functional Food Ingredients: Structure, Function and Health Benefits)
►▼
Show Figures
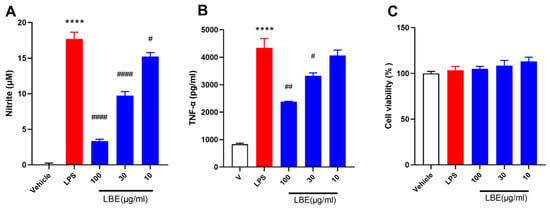
Figure 1
Open AccessArticle
Cholestane-3β,5α,6β-triol Induces Multiple Cell Death in A549 Cells via ER Stress and Autophagy Activation
by
Jiaxi Chen, Jieping Zhang, Lijuan Cai, Li Guo, Zhenyu Cai, Hua Han and Wen Zhang
Mar. Drugs 2024, 22(4), 174; https://doi.org/10.3390/md22040174 - 13 Apr 2024
Abstract
Cholestane-3β,5α,6β-triol (CT) and its analogues are abundant in natural sources and are reported to demonstrate cytotoxicity toward different kinds of tumor cells without a deep probe into their mechanism of action. CT is also one of the major metabolic oxysterols of cholesterol in
[...] Read more.
Cholestane-3β,5α,6β-triol (CT) and its analogues are abundant in natural sources and are reported to demonstrate cytotoxicity toward different kinds of tumor cells without a deep probe into their mechanism of action. CT is also one of the major metabolic oxysterols of cholesterol in mammals and is found to accumulate in various diseases. An extensive exploration of the biological roles of CT over the past few decades has established its identity as an apoptosis inducer. In this study, the effects of CT on A549 cell death were investigated through cell viability assays. RNA-sequencing analysis and western blot of CT-treated A549 cells revealed the role of CT in inducing endoplasmic reticulum (ER) stress response and enhancing autophagy flux, suggesting a putative mechanism of CT-induced cell-death activation involving reactive oxygen species (ROS)-mediated ER stress and autophagy. It is reported for the first time that the upregulation of autophagy induced by CT can serve as a cellular cytotoxicity response in accelerating CT-induced cell death in A549 cells. This research provides evidence for the effect of CT as an oxysterol in cell response to oxidative damage and allows for a deep understanding of cholesterol in its response in an oxidative stress environment that commonly occurs in the progression of various diseases.
Full article
(This article belongs to the Section Marine Pharmacology)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Structural Insights into the Marine Alkaloid Discorhabdin G as a Scaffold towards New Acetylcholinesterase Inhibitors
by
Andrea Defant, Giacomo Carloni, Nicole Innocenti, Tomaž Trobec, Robert Frangež, Kristina Sepčić and Ines Mancini
Mar. Drugs 2024, 22(4), 173; https://doi.org/10.3390/md22040173 - 12 Apr 2024
Abstract
In this study, Antarctic Latrunculia sponge-derived discorhabdin G was considered a hit for developing potential lead compounds acting as cholinesterase inhibitors. The hypothesis on the pharmacophore moiety suggested through molecular docking allowed us to simplify the structure of the metabolite. ADME prediction and
[...] Read more.
In this study, Antarctic Latrunculia sponge-derived discorhabdin G was considered a hit for developing potential lead compounds acting as cholinesterase inhibitors. The hypothesis on the pharmacophore moiety suggested through molecular docking allowed us to simplify the structure of the metabolite. ADME prediction and drug-likeness consideration provided valuable support in selecting 5-methyl-2H-benzo[h]imidazo[1,5,4-de]quinoxalin-7(3H)-one as a candidate molecule. It was synthesized in a four-step sequence starting from 2,3-dichloronaphthalene-1,4-dione and evaluated as an inhibitor of electric eel acetylcholinesterase (eeAChE), human recombinant AChE (hAChE), and horse serum butyrylcholinesterase (BChE), together with other analogs obtained by the same synthesis. The candidate molecule showed a slightly lower inhibitory potential against eeAChE but better inhibitory activity against hAChE than discorhabdin G, with a higher selectivity for AChEs than for BChE. It acted as a reversible competitive inhibitor, as previously observed for the natural alkaloid. The findings from the in vitro assay were relatively consistent with the data available from the AutoDock Vina and Protein-Ligand ANTSystem (PLANTS) calculations.
Full article
(This article belongs to the Special Issue Marine Drug Discovery through Molecular Docking)
►▼
Show Figures
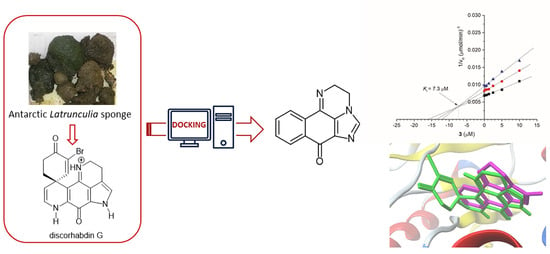
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Marine Drugs Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal Browser-
arrow_forward_ios
Forthcoming issue
arrow_forward_ios Current issue - Vol. 22 (2024)
- Vol. 21 (2023)
- Vol. 20 (2022)
- Vol. 19 (2021)
- Vol. 18 (2020)
- Vol. 17 (2019)
- Vol. 16 (2018)
- Vol. 15 (2017)
- Vol. 14 (2016)
- Vol. 13 (2015)
- Vol. 12 (2014)
- Vol. 11 (2013)
- Vol. 10 (2012)
- Vol. 9 (2011)
- Vol. 8 (2010)
- Vol. 7 (2009)
- Vol. 6 (2008)
- Vol. 5 (2007)
- Vol. 4 (2006)
- Vol. 3 (2005)
- Vol. 2 (2004)
- Vol. 1 (2003)
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Applied Biosciences, JMSE, Marine Drugs, Toxins, Molecules
Marine Biotoxins and Bioactive Marine Natural Products
Topic Editors: Naomasa Oshiro, Ana Gago-Martínez, Takeshi Tsumuraya, Tsuyoshi IkeharaDeadline: 31 August 2024
Topic in
Molecules, Pharmaceutics, Antibiotics, Microorganisms, Biomolecules, Marine Drugs, Polymers, IJMS
Antimicrobial Agents and Nanomaterials
Topic Editors: Sandra Pinto, Vasco D. B. BonifácioDeadline: 30 September 2024
Topic in
Biomolecules, IJMS, Molecules, Sci. Pharm., Marine Drugs, Plants
Antioxidant Activity of Natural Products, 2nd Volume
Topic Editors: José Virgílio Santulhão Pinela, Maria Inês Moreira Figueiredo Dias, Carla Susana Correia Pereira, Alexandra PlácidoDeadline: 30 September 2025

Conferences
Special Issues
Special Issue in
Marine Drugs
Characterization of Structure-Activity Relationships in Marine Natural Products
Guest Editors: Carmen Festa, Simona De MarinoDeadline: 30 April 2024
Special Issue in
Marine Drugs
Sustainable Production and Cultivation Systems for Coastal and Marine Organisms Targeting the Extraction of Bioactive Compounds
Guest Editors: Catarina Guerreiro Pereira, Luísa Custódio, Maria João RodriguesDeadline: 15 May 2024
Special Issue in
Marine Drugs
Biosynthesis, Metabolism, Pharmacology and Biological Receptors of Marine Algal Toxins
Guest Editor: Kathleen ReinDeadline: 31 May 2024
Special Issue in
Marine Drugs
Marine Bacteriophages and Their Applications
Guest Editor: Ipek KurtbokeDeadline: 15 June 2024
Topical Collections
Topical Collection in
Marine Drugs
Microalgal Active Biomolecules
Collection Editor: Cédric Delattre
Topical Collection in
Marine Drugs
Marine Compounds and Cancer
Collection Editors: Friedemann Honecker, Sergey A. Dyshlovoy
Topical Collection in
Marine Drugs
Papers from “Sino–Italian Symposium on Bioactive Natural Products”
Collection Editors: Orazio Taglialatela-Scafati, Hong Wang